
Concept explainers
MATHEMATICAL The enzyme
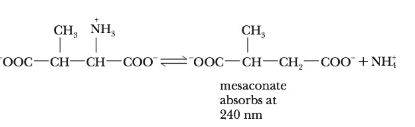
[V. Williams and J. Selbin, J. Biol. Chem. 239, 1636 (1964)]. The
Interpretation:
The
Concept introduction:
In an enzymatic reaction,
To determine
To draw this plot, substrate concentration
The
Answer to Problem 28RE
Plotting the data gives the following straight line:
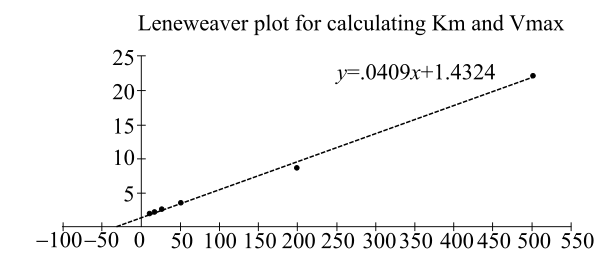
From this straight-line graph, the value of
Explanation of Solution
Given information:
Following data containing substrate concentrations and velocity of the reaction at that substrate concentration, is given.
In enzyme kinetics, to determine
But
Modification of the equation is
This equation can be plotted on the graph,
The enzyme, b-methyl aspartate, catalyzes the deamination of b-methyl aspartate. By plotting the given data into a Lineweaver–Burk plot, Km for this reaction is 2.86 × 10-2 mole. Although in this case, concentration cannot be determined directly. Absorbance values were used instead, as a matter of convenience.
Want to see more full solutions like this?
Chapter 6 Solutions
Bundle: Biochemistry, Loose-leaf Version, 9th + Lms Integrated For Owlv2, 1 Term Printed Access Card
- REFLECT AND APPLY A model is proposed to explain the reaction catalyzed by an enzyme. Experimentally obtained rate data fit the model to within experimental error. Do these findings prove the model?arrow_forwardREFLECT AND APPLY Noncompetitive inhibition is a limiting case in which the effect of binding inhibitor has no effect on the affinity for the substrate and vice versa. Suggest what a LineweaverBurk plot would look like for an inhibitor that had a reaction scheme similar to that on page 159 (noncompetitive inhibition reaction), but where binding inhibitor lowered the affinity of EI for the substrate.arrow_forwardREFLECT AND APPLY When we compare the binding of I and of S to the enzyme in a mixed noncompetitive inhibitor, we assumed that the binding of I decreased the affinity of the enzyme for S. What would happen if the opposite were true?arrow_forward
- REFLECT AND APPLY What is the relationship between a transition-state analog and the induced-fit model of enzyme kinetics?arrow_forwardREFLECT AND APPLY Would nature rely on the same enzyme to catalyze a reaction either way (forward or backward) if the DG were 0.8kcalmol1? If it were 5.3kcalmol1?arrow_forwardMATHEMATICAL If a reaction can be written AB, and the G is 20kJmol1, what would the substrate/product ratio have to be for the reaction to be thermodynamically favorable?arrow_forward
- REFLECT AND APPLY What are some of the difficulties in determining the exact number of protons pumped across the inner mitochondrial membrane by the respiratory complexes?arrow_forwardMATHEMATICAL For an enzyme that displays MichaelisMenten kinetics, what is the reaction velocity, V (as a percentage of Vmax), observed at the following values? (a) [S]=KM (b) [S]=0.5KM (c) [S]=0.1KM (d) [S]=2KM (e) [S]=10KMarrow_forwardMATHEMATICAL Consider the reaction AB+C, where G=0.00. (a) What is the value of G (not G) when the initial concentrations of A, B, and C are 1 M, 103M,and106M? (b) Try the same calculations for the reaction D+EF, for the same relative order of concentrations. (c) Try the same calculations for the reaction GH, if the concentrations are 1Mand103M for G and H, respectively.arrow_forward
- REFLECT AND APPLY Would you expect an irreversible inhibitor of an enzyme to be bound by covalent or by non-covalent interactions? Why?arrow_forwardREFLECT AND APPLY Suggest a reason why heating a solution containing an enzyme markedly decreases its activity. Why is the decrease of activity frequently much less when the solution contains high concentrations of the substrate?arrow_forwardREFLECT AND APPLY You are studying with a friend who is describing the Bohr effect. She tells you that in the lungs, hemoglobin binds oxygen and releases hydrogen ion; as a result, the pH in- creases. She goes on to say that in actively metabolizing muscle tissue, hemoglobin releases oxygen and binds hydrogen ion and, as a result, the pH decreases. Do you agree with her reasoning? Why or why not?arrow_forward
 BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
