
CHEM: STRUC & PROP (LL) VOL 2 >C PKG<
2nd Edition
ISBN: 9780137390922
Author: Tro
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 6, Problem 36E
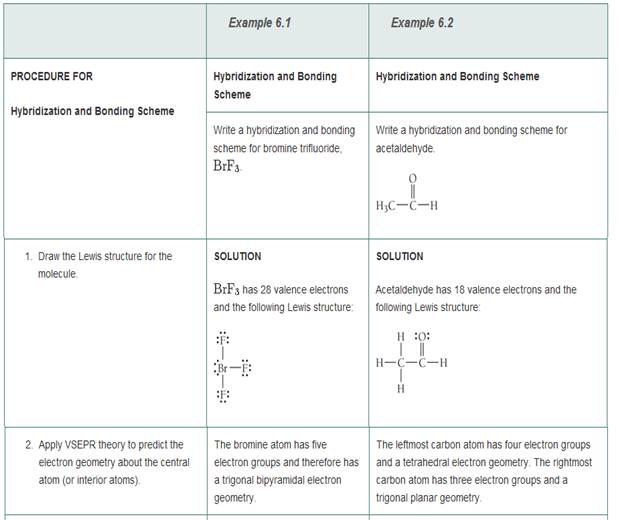
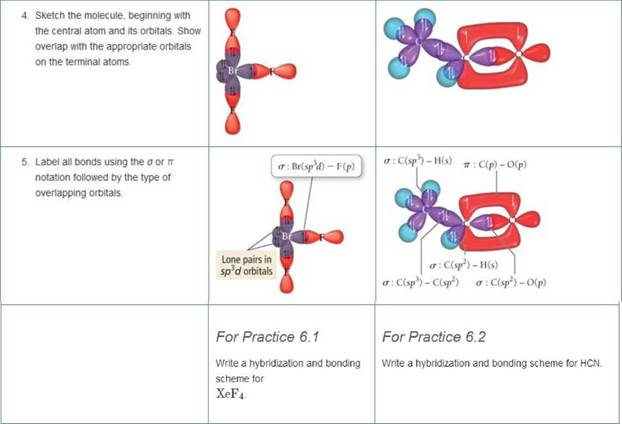
Write a hybridization and bonding scheme for each molecule or ion. Sketch the structure, including overlapping orbitals, and label all bonds using the notation shown in Examples 6.1 and 6.2
- SO32-
- PF6-
- BrF3
- HCN

Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The molecular structure of the molecule sulfur cyanide trifluoride, CF3NS, is characterized by the following bond lengths: C-N/ 116 picometers, S-C/ 174 picometers, and S-F/ 160 picometers. Using the observed bond lengths as a guide, draw a realistic lewis structure for sulfur cyanide tirfluoride and assign formal charges to each atom. Are there any resonance structures? Predict the hybridization of the C , N, and S atoms.
Consider the following ion: BrO3−.
What is the electronic geometry of BrO3−? What is its molecular shape?
e) Does BrO3− have a dipole moment? Briefly justify your answer.
f) On average, would you expect IO3− to have longer or shorter bonds than BrO3−?
Briefly explain your answer.
The most stable forms of the nonmetals in groups 4A, 5A, and 6A of the first period are molecules with multiple bonds. Beginning with the second period, the most stable forms of the nonmetals of these groups are molecules without multiple bonds. Propose an explanation for this observation based on valence bond theory.
Chapter 6 Solutions
CHEM: STRUC & PROP (LL) VOL 2 >C PKG<
Ch. 6 - Prob. 1ECh. 6 - What is a chemical bond according to valence bond...Ch. 6 - In valence bond theory, what determines the...Ch. 6 - In valence bond theory, the interaction energy...Ch. 6 - What is hybridization? Why is hybridization...Ch. 6 - How does hybridization of the atomic orbitals in...Ch. 6 - How is the number of hybrid orbitals related to...Ch. 6 - Sketch each hybrid orbital sp sp2 sp3 sp3d sp3d2Ch. 6 - Prob. 9ECh. 6 - Name the hybridization scheme that corresponds to...
Ch. 6 - What is a chemical bond according to molecular...Ch. 6 - Explain the difference between hybrid atomic...Ch. 6 - What is a bonding molecular orbital?Ch. 6 - Prob. 14ECh. 6 - What is the role of wave interference in...Ch. 6 - Prob. 16ECh. 6 - Prob. 17ECh. 6 - Prob. 18ECh. 6 - Prob. 19ECh. 6 - Prob. 20ECh. 6 - Prob. 21ECh. 6 - When applying molecular orbital theory to...Ch. 6 - In molecular orbital theory, what is a nonbonding...Ch. 6 - Write a short paragraph describing chemical...Ch. 6 - The valence electron configurations of several...Ch. 6 - The valence electron configurations of several...Ch. 6 - Draw orbital diagrams (boxes with arrows in them)...Ch. 6 - Draw orbital diagrams (boxes with arrows in them)...Ch. 6 - Prob. 29ECh. 6 - Draw orbital diagrams (boxes with arrows in them)...Ch. 6 - Which hybridization scheme allows the formation of...Ch. 6 - Which hybridization scheme allows the central atom...Ch. 6 - Write a hybridization and bonding scheme for each...Ch. 6 - Write a hybridization and bonding scheme for each...Ch. 6 - Write a hybridization and bonding scheme for each...Ch. 6 - Write a hybridization and bonding scheme for each...Ch. 6 - Write a hybridization and bonding scheme for each...Ch. 6 - Write a hybridization and bonding scheme for each...Ch. 6 - Consider the structure of the amino acid alanine...Ch. 6 - Consider the structure of the amino acid aspartic...Ch. 6 - Sketch the bonding molecular orbital that results...Ch. 6 - Sketch the antibonding molecular orbital that...Ch. 6 - Draw an MO energy diagram and predict the bond...Ch. 6 - Draw an MO energy diagram and predict the bond...Ch. 6 - Sketch the bonding and antibonding molecular...Ch. 6 - Sketch the bonding and antibonding molecular...Ch. 6 - Using the molecular orbital energy ordenng for...Ch. 6 - Using the molecular orbital energy ordering for...Ch. 6 - Apply molecular orbital theory to predict if each...Ch. 6 - Apply molecular orbital theory to predict if each...Ch. 6 - According to MO theory, which molecule or ion has...Ch. 6 - According to MO theory, which molecule or ion has...Ch. 6 - Draw an MO energy diagram for CO. (Use the energy...Ch. 6 - Draw an MO energy diagram for HCI. Predict the...Ch. 6 - For each compound, draw the Lewis structure,...Ch. 6 - For each compound, draw the Lewis structure,...Ch. 6 - Amino acids are biological compounds that link...Ch. 6 - The genetic code is based on four different bases...Ch. 6 - The structure of caffeine, present in coffee and...Ch. 6 - The structure of acetylsalicylic acid (aspirin) is...Ch. 6 - Draw a molecular orbital energy diagram for CIF....Ch. 6 - Draw Lewis structures and MO diagrams for CN+, CN,...Ch. 6 - Bromine can form compounds or ions with any number...Ch. 6 - The compound C3H4 has two double bonds. Describe...Ch. 6 - How many hybrid orbitals do we use to describe...Ch. 6 - Prob. 66ECh. 6 - In VSEPR theory, which uses the Lewis model to...Ch. 6 - The resuts of a molecular orbital calculation for...Ch. 6 - Prob. 69ECh. 6 - cis-2-Butene isomerizes (changes its structure) to...Ch. 6 - The ion CH5 + can form under very special...Ch. 6 - Neither the VSEPR model nor the hybridization...Ch. 6 - Prob. 73ECh. 6 - The most stable forms of the nonmetals in groups...Ch. 6 - Consider the bond energies of three iodine...Ch. 6 - How many atomic orbitals form a set of sp3hybrid...Ch. 6 - Have each group member pick one of these...Ch. 6 - Divide your group into two subgroups. Have one...Ch. 6 - A molecular orbital calculation for Hi results in...Ch. 6 - Determine the hybridization about 0 in CH3OH.Ch. 6 - Determine the hybridization about C in H2CO.Ch. 6 - According to the valance bond theory, which kind...Ch. 6 - Use molecular orbital theory to determine the bond...Ch. 6 - Use molecular orbital theory to predict which...Ch. 6 - Use molecular orbital theory to determine which...Ch. 6 - Which hybridization scheme occurs about nitrogen...Ch. 6 - Prob. 8SAQCh. 6 - Prob. 9SAQCh. 6 - Prob. 10SAQCh. 6 - Which type of orbitals overlap to form the sigma...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What are the relationships among bond order, bond energy, and bond length? Which of these quantities can be measured?arrow_forwardIt is possible to write a simple Lewis structure for the SO42- ion, involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs. (a) Draw the two Lewis structures. (b) What geometries are predicted for the two structures? (c) What is the hybridization of sulfur in each case? (d) What are the formal charges of the atoms in the two structures?arrow_forward• identify sigma and pi bonds in a molecule and explain the difference between them.arrow_forward
- The XeO3 molecule displays approximately which bond angles (in degrees)about the central Xe? VSEPR theory predicts which molecular structure (arrangement of bonded atoms about the central Xe atom) for XeO3?arrow_forwardDescribe what makes up a pi, π, bond including the orbitals involved and how they overlap.arrow_forward4. Predict and show the overall molecular geometry/shape and the molecular geometry/shape of the BrCl4 anion. 5. Given the following Lewis structure for the azide anion, N3, calculate the formal charges for the three N atoms and display them over the Natoms in the structure. [:N_N=N:] 6. State the expected hybridization scheme of the S atom in the SOF4+ cation.arrow_forward
- Draw the Lewis structure of CH₃CCH and then choose the appropriate set of hybridization states for the three central atoms. Your answer choice is independent of the orientation of your drawn structure.arrow_forwardIdentify and draw the Lewis structure. Label each bond with σ and π, and give the hybridization of each bondarrow_forwardConstruct the molecular orbital diagram for SrCl. Would yo u expect the bond length of SrCl+ to be longer or shorter than that of SrCl? Explain and elaborate.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Linear Combination of Atomic Orbitals LCAO; Author: Edmerls;https://www.youtube.com/watch?v=nq1zwrAIr4c;License: Standard YouTube License, CC-BY
Quantum Molecular Orbital Theory (PChem Lecture: LCAO and gerade ungerade orbitals); Author: Prof Melko;https://www.youtube.com/watch?v=l59CGEstSGU;License: Standard YouTube License, CC-BY