
Concept explainers
Slope Field In Exercises 67-70, (a) sketch an approximate solution of the differential equation satisfying the initial condition by hand on the slope field, (b) find the particular solution that satisfies the initial condition, and (c) use a graphing utility to graph the particular solution. Compare the graph with the hand-drawn graph in part (a). To print an enlarged copy of the graph, go to MathGraphs.com.
Differential Equation Initial Condition
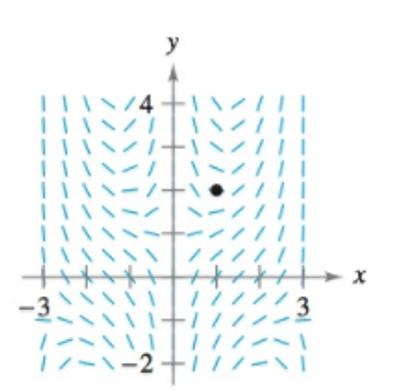
Figure for 69
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Calculus: Early Transcendental Functions (MindTap Course List)
- Van der Waals Equation In Exercise 18 at the end of Section 2.3, we discussed the ideal gas law, which shows the relationship among volume V, pressure p, and temperature T for a fixed amount 1 mole of a gas. But chemists believe that in many situations, the van der Waals equation gives more accurate results. If we measure temperature T in kelvins, volume V in liters, and pressure p in atmosphere 1 atm is the pressure exerted by the atmosphere at sea level, then the relationship for carbon dioxide is given by p=0.082TV0.0433.592V2atm What volume does this equation predict for 1 mole of carbon dioxide at 500 kelvins and 100 atm?Suggestion: Consider volumes ranging from 0.1 to 1 liter.arrow_forwardUse a graphing utility to graph the slope field for the differential equation, (a) find the particular solutions of the differential equation passing through the given points, and (b) use a graphing utility to graph the particular solutions on the slope field.arrow_forwardApplication of First Order linear differential equation show complete solutionarrow_forward
 Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning
Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning
