
(a)
Interpretation:
Using the recommended daily dietary intake of calcium the amount of calcium required for adult men and premenopausal women should be identified.
Concept Introduction:
Importance of Calcium in diet:
- 1. Calcium is required by human body for muscle contraction, hormones release and transmission of messages via nerves.
- 2. Correct intake of calcium helps one from osteoporosis which occurs as density of the bone decreases and results in structural defect and weakness of bones.
(b)
Interpretation:
The mass of calcium citrate required to attain the recommended daily intake of calcium should be calculated.
Concept Introduction:
Calcium citrate: It is recommended for calcium deficiency diseases namely rickets, tetany and osteoporosis. The molecular formula of Calcium citrate is
Importance of Calcium in diet:
- 1. Calcium is required by human body for muscle contraction, hormones release and transmission of messages via nerves.
- 2. Correct intake of calcium helps one from osteoporosis which occurs as density of the bone decreases and results in structural defect and weakness of bones.
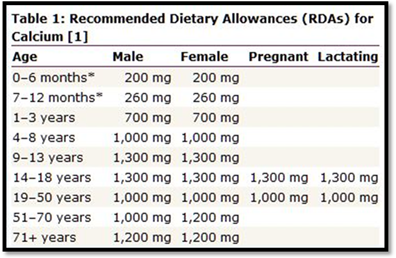
The table that depicts recommended daily intake of calcium is as follows,
Moles: One mole is equivalent to the mass of the substance consists same number of units equal to the atoms present in
The mole is actually quantity of particles that is
The sum of mass of all atoms present in formula of chemical substance is referred as molecular or formula weight of that substance.
Mass: It is the quantitative measure of a substance. The amount of matter present in substance is expressed as mass. The
Molar mass: It is obtained by dividing the mass of substance with the amount of substance and the S.I. unit of molar mass is
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
FUND.OF GEN CHEM CHAP 1-13 W/ACCESS
- How many milliliters of Children’s Motrin (100 mg of ibuprofen per 5 mL) are needed to give a child a dose of 60 mg?arrow_forwardWhat is the molar mass of diazepam (Valium), C16H13ClN2O?arrow_forwardIf a pediatric vitamin contains 1500 units of vitamin A per milliliter of solution, how many units of vitamin A would be administered to a child given 2 drops of a solution from a drpper calibrated to deliver 20 drops per milliar of solution?arrow_forward
- Propanamide and methyl acetate have about the same molar mass, both are quite soluble in water, and yet the boiling point of propanamide is 486 K, whereas that of methyl acetate is 330 K. Explain.arrow_forwardWrite the Structure Activity Relationship (SAR) of Barbiturates? Please write at your own words.arrow_forwardWhy is it difficult to determine an exact number for P/O ratios?arrow_forward
- If glucose, phosphate, and glucose-6-phosphate arecombined in concentrations of 4.8, 4.8, and 0.25 mM,respectively, what is the equilibrium constant for thehydrolysis of glucose-6-phosphate at a temprature of258C?arrow_forwardif you have Quinoa Serving size -170 grams Carbohydrates – 109 grams calculate the DV % for these values?arrow_forwardWith a stock concentration of 0.2817 g/L, determine each standard's concentrations. What is the mass of iron in the vitamin?arrow_forward
- Calculate the molar mass for each of the followingarrow_forwardCreatinine, C4H7N3O, is a by-product of muscle metabolism, and creatinine levels in the body are known to be a fairly reliable indicator of kidney function. The normal level of creatinine in the blood for adults is approximately 1.0 mg per deciliter (dL) of blood. If the density of blood is 1.025 g/mL, calculate the molarity of a normal creatinine level in a 10.0 mL blood sample.arrow_forwardConsider the following blood group A, B, AB and O.What are the sugar(carbohydrate ) content of their glycolipid?arrow_forward
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning





