
Interpretation:
The ground state electronic configurations of the given elements are to be obtained.
Concept introduction:
Aufbau Principle states that every electron occupies the orbitals in the increasing order of their energy.
The four steps for writing the ground state electronic configuration of an element are shown below:
An electron enters the lowest energy orbital.
A maximum of two electrons can occupy each orbital.
In a degenerate orbital, the electrons do not pair until all the orbitals are occupied with at least one electron.
Electrons occupy the orbitals in the following order:
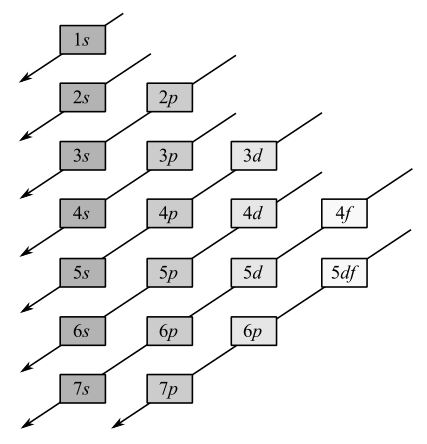
Noble gas core is used to represent electron configuration of all the elements except hydrogen and helium.
Noble gas core is an abbreviation in an atom’s electron configuration. It replaces the electron configuration of noble gas element most recently precede the element.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Looseleaf Study Guide For Chemistry
- The successive ionization energies for an unknown element are I1 = 896 kJ/mol I2 = 1752 kJ/mol I3 = 14,807 kJ/mol I4 = 17,948 kJ/mol To which family in the periodic table does the unknown element most likely belong?arrow_forwardOrder the atoms in each of the following sets from the least negative electron affinity to the most. a. N, O, F b. Al, Si, Parrow_forwardWhat is the maximum number of electrons that can be identified with each of the following sets of quantum numbers? In some cases, the answer may be none In such cases, explain why none is the correct answer. (a) n = 4, = 3, m = 1 (b) n = 6, = 1, m = 1, ms = 1/2 (c) n= 3, = 3, m = 3arrow_forward
- • identify an orbital (as 1s, 3p, etc.) from its quantum numbers, or vice versa.arrow_forward6.49 Which of these electron configurations are for atoms in the ground state? In excited states? Which are impossible? (a) 1s22s1, (b) 1s22s22p3, (c) [Ne] 3s23p34s1, (d) [Ne] 3s23p64s23d2, (e) [Ne] 3s23p64f 4, (f) 1s22s22p43s2arrow_forwardWhat is the maximum number of electrons that can occupy a f subshell (l = 3)?arrow_forward
- The energy emitted when an electron moves from a higher energy state to a lower energy state in any atom can be observed as electromagnetic radiation. (a) Which involves the emission of less energy in the H atom, an electron moving from n = 4 to n = 2 or an electron moving from n = 3 to n = 2? (b) Which involves the emission of more energy in the H atom, an electron moving from n = 4 to n = 1 or an electron moving from n = 5 to n = 2? Explain fully.arrow_forwardUsing orbital box diagrams, depict an electron configuration for each of the following ions: (a) Mg2+, (b) K+, (c) Cl, and (d) O2.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning  Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





