
Interpretation:
Pathway for finding “moles of B” from “grams of A” using the conversion factors has to be chosen from the given options.
Concept Introduction:
Using the

Molar mass of a substance gives a relationship between the number of grams and number of moles of that particular substance.

The Chemical formula subscript also provides relationship between the number of moles of substance to the number of moles of its individual components present in the substance.

The moles of compound are related to moles of another compound by the coefficients present in the balanced chemical equation.
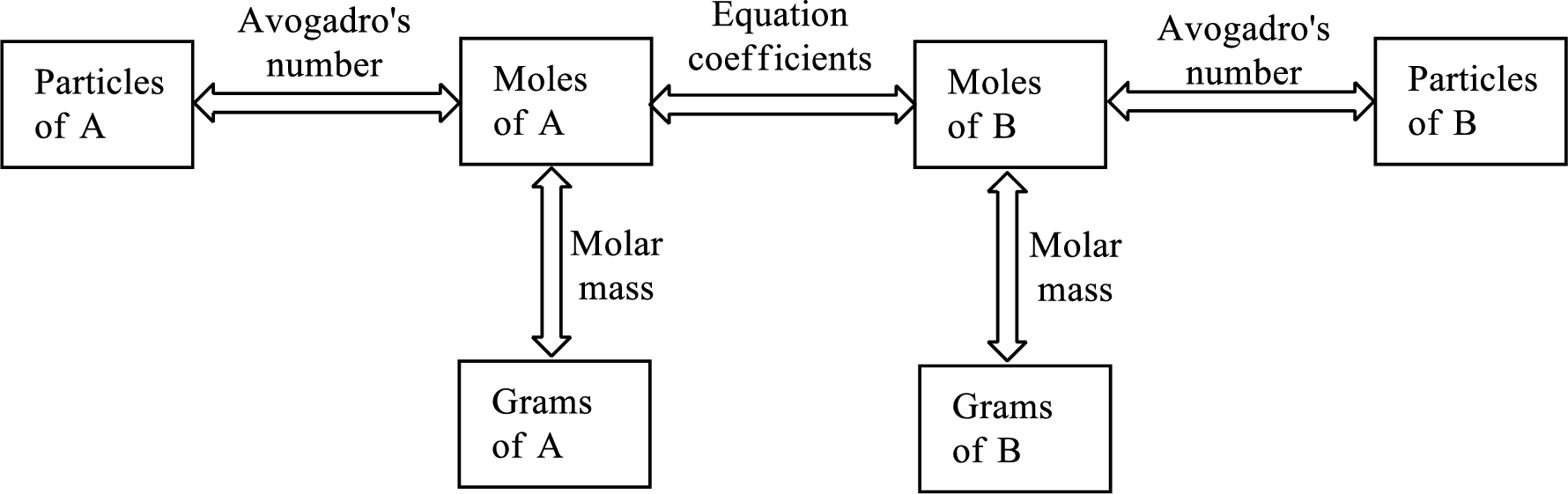
All the above relationship can be put into a single diagram as shown below,
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- rug-B has an elimination half-life of 9 hours, an absorption half-life of 20 minutes and linear harmacokinetics. In a patient, the plasma concentration five hours from the administration of two 60 mg cablets of the drug is 8 ug/mL. What would you expect to be the plasma concentration five hours from the administration of one 60 mg tablets? 1 ug/mL Answer 1-1arrow_forwardA KCC student titrated 100.00 mL of a NaOH solution using 55.00 mL of a 0.1000M H2SO4 solution. How many moles of acid were used ? This is part 1 of a titration problem H2SO4 + 2 NAOH Na2SO4 + 2 H20 O A. 0.0055 mol B. 0.0100 mol O C. 0.100 mol D. 0.550 mol O E. 5.50 molarrow_forwardUsing conversion factors, solve each of the following clinicalproblems:a. A nurse practitioner prepares an injection of promethazine,an antihistamine used to treat allergic rhinitis. If thestock bottle is labeled 25 mg /mL and the order is a doseof 12.5 mg, how many milliliters will the nurse draw up inthe syringe?b. You are to give ampicillin 25 mg/kg to a child with a massof 62 lb. If stock on hand is 250 mg /capsule, how manycapsules should be given?arrow_forward
- A client has been ordered Minipress (prazosin hydrochloride) 2 mg po twice a day (first 2 mg dose to be taken in the morning and the second 2 mg dose to be taken at bedtime). The medication is available as Minipress (prazosin hydrochloride) 1 mg capsules. 1. what is the main conversions factor that you will be using to solve this equation? 2. How many capsules should be administered to this client for the whole day?arrow_forwardHow do you create a graph like the one in the picture? Here is the data from my lab. Time Concentration Mean 1 hr 100 4.7 70 6.6 50 6 30 4.2 10 0.96 1 0.08 0.1 0 control 0.08 24 hr 100 10 70 10 50 10 30 10 10 10 1 3.7 0.1 1.5 control 0.79 48 hr 100 10 70 10 50 10 30 10 10 10 1 6.96 0.1 3.4 control 1.8 A tutorial would be much appreciated or even a video that walks you through the process. Cheersarrow_forwardYou have an order for 1 gram of Cefazolin in D5W 100 ml. You have added 5 ml of sterile water to the 1 gram vial to reconstitute powder. However the recommended manufacturer’s diluent amount is 10 ml of sterile water for a final concentration of 100 mg/ml. How would reconstituting the vial with 5 mls affect the concentration and the final calculated dose. Please answer with explanation ASAP. I will really upvote. Thanksarrow_forward
- In the experiment performed for the determination of cholesterol, the function of the calibration curve obtained using 25-400mg/dL standard cholesterol solutions was determined as y= 0.0009x. If the amount of cholesterol in the serum sample is determined as 3.11 g/L, what is the absorbance value of the sample? A. 0,280 B. 0.028 C. 0.208 D. 0.82arrow_forwardThe dose level of Drug XYZ is 60 mg/m2 (Hint: Use the table found in p.19 for the conversion factors)a. What is the dose in body weight (mg/kg) basis in dogs?b. What is the dose in body weight (mg/kg) basis in rats?c. What is the dose in body weight (mg/kg) basis in monkeys?d. What is the dose in body weight (mg/kg) basis in humans? Reference:Guidance for IndustryEstimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers the picture attached to my question is page 19 on my pdfarrow_forwardWhat is the approximate rate of change of A340 measured? i.e. ΔA340 / min = __________ What rate of change of A340 would you predict if 30 ml of the ADH solution was tested in the same way (i.e. half the amount of protein) ? i.e. ΔA340 / min = __________ What rate of change of A340 would you predict if 60 ml of a 0.5 μM ADH solution was tested in the same way? i.e. ΔA340 / min = __________ As well as writing your answers, explain your reasoning.arrow_forward
- What is the purpose of checking the %GC in our NGS data? What is the ideal %GC value? Explain why this is the ideal value.arrow_forward5. Using this formula Cu = Au x Cstd. %3D Astd Where: C = concentration A = absorbance U = unknown/sample STD = standard Compute for the glucose concentration of the patient's sample. Given: Absorbance of the standard = 0.075; Concentration of the standard = 10.54 Absorbance = 0.083 Convert your answer to mmol/L or S.I. using the conversion factor for glucose which is 0.055 Please show your computation.arrow_forwardA particular drug has first-order elimination rate kinetics and follows a one-compartment model. Which of the following plots would you expect to result in a curved line? A. plasma concentration versus time B. natural log of plasma concentration versus time ( for pharmaceutical formulation module)arrow_forward
 Cardiopulmonary Anatomy & PhysiologyBiologyISBN:9781337794909Author:Des Jardins, Terry.Publisher:Cengage Learning,
Cardiopulmonary Anatomy & PhysiologyBiologyISBN:9781337794909Author:Des Jardins, Terry.Publisher:Cengage Learning,


