
Concept explainers
a)
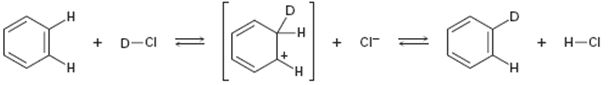
Interpretation:
Curved arrows are to be added to the polar reaction given to indicate the flow of electrons.
Concept introduction:
Curved arrows start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained both in the source and sink.
To add:
Curved arrows to indicate the flow of electrons in the polar reaction given.
b)
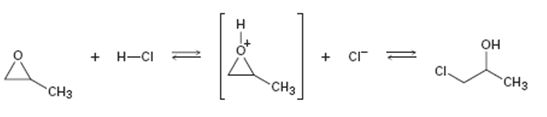
Interpretation:
Curved arrows are to be added to the polar reaction given to indicate the flow of electrons.
Concept introduction:
Curved arrows start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained both in the source and sink.
To add:
Curved arrows to indicate the flow of electrons in the polar reaction given.
Trending nowThis is a popular solution!

Chapter 6 Solutions
Organic Chemistry - Owlv2 Access (4 Term)
- For each of the following compounds and ions,1. Draw a Lewis structure.2. Show the kinds of orbitals that overlap to form each bond.3. Give approximate bond angles around each atom except hydrogen.(a) [NH2]- (b) [CH2OH]+ (c) CH2“N¬CH3(d) CH3¬CH“CH2 (e) HC‚C¬CHO (f) H2N¬CH2¬CNarrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided resonance structures, draw the curved electron-pushing arrows to show the interconversion between resonance hybrid contributors. Be sure to account for all bond-breaking and bond-making steps. Drawing Arrows N ▸ Qarrow_forwardComplete the equation for the reaction between the following Lewis acid-base pair. Use curved arrows to show the flow of electrons in the reaction and draw the product. Assign lone pairs and radical electrons where appropriate. Apply formal charges where appropriate. • Draw the appropriate electron-flow arrows. • Use the "starting points" menu to revert to the original molecule(s) shown. • Omit + signs between structures. ● / CH3 1- H₂C-C CH3 H در St ? ChemDoodleⓇarrow_forward
- Which sets of curved arrows accounts for the protonation of propene with HI?arrow_forwardConsider the acid-base reaction below to answer the following question(s). + H-Q-H - CH₂ base acid A) Using the curved arrow formalism, show the flow of electrons for this reaction. B)Write the products of this reaction. CH,CH-arrow_forwardDraw thee other resonance structures for the following carbocation. Use electronpushing arrows in order to derive one resonance structure from another. Indicate theformal charge on each atom in your structures.arrow_forward
- Select the single best answer. Considering only electron density, will the following reaction occur? CI CH30: + yes noarrow_forwardIn the following acid - base reactions, a) Draw Lewis structures of the reactants and the products. b) Determine which species are acting as electrophiles (acids) and which are acting as nucleophiles (bases). c) Use the curved - arrow formalism to show the movement of electron pairs in these reactions and the imaginary movement in the resonance hybrids of the products. d) Indicate which reactions are best termed Brønsted-Lowry acid - base reactions i. CH3CHO + HCI-- > CH3CH2O + + Cl- ii. CH3CHO + OH- - - > CH3CO-(OH) Harrow_forwardComplete the equation for the following proton-transfer reaction by using curved arrows to show the flow of electron pairs and drawing the products of the reaction. • Draw all atoms, including hydrogen atoms. Apply formal charges where appropriate. Assign lone pairs and radical electrons where appropriate. • Use the "starting points" menu to revert to the original molecule(s) shown. • Draw the appropriate electron-flow arrows. • Omit + signs between structures. ● ● H-O: == starting points == ↑ TAYY H T H-C-H H-N-H T H در ? ChemDoodlearrow_forward
- which of the following structures is different from the other three?arrow_forwardComplete the equation for the following proton-transfer reaction by using curved arrows to show the flow of electron pairs and drawing the products of the reaction. • Draw all atoms, including hydrogen atoms. • Apply formal charges where appropriate. Assign lone pairs and radical electrons where appropriate. • Use the "starting points" menu to revert to the original molecule(s) shown. • Draw the appropriate electron-flow arrows. • Omit + signs between structures. 0▾ HH HH == starting points == V H-CI: کر ChemDoodle ?arrow_forwardWrite an equation for the reaction of CH3 C(=O)OCH₂ CH3 with BF3, a Lewis acid, and show by the use of curved arrows how the reaction occurs. Show all hydrogen atoms that are not attached to a carbon atom. Apply formal charges where appropriate. • Assign lone pairs and radical electrons where appropriate. ● Use the "starting points" menu to revert to the original molecule(s) shown. • Draw the appropriate electron-flow arrows. Omit+ signs between structures. ● CH3 c=0 :0: d HC ។ CH3 O n ChemDoodle Previous Nextarrow_forward
