
Concept explainers
Interpretation:
The IUPAC names of each of the given compounds are to be given by using
Concept introduction:
The priority of groups in nomenclature is assigned according to Cahn-Ingold-Prelog (CIP Rule) convention rules:
The higher the
If priority cannot be assigned according to atomic mass, then assign the priority according to first point of difference.
If both the priority groups are on the same side of double-bonded carbon atom, then it is known as
But, if both the priority groups are diagonal to each other, then it is
Rule for R/S Nomenclature:
4B(+)3B(-)2B(+)1B(-)
4B(+) means if 4th priority group is below then from 1 to 2 to 3 if leads a clockwise rotation then its R and if anticlockwise then it is S.
3B(-) means if 3rd priority group is below then from 1 to 2 to 4 if leads a clockwise rotation then its S and if anticlockwise then it is R.
Answer to Problem 1PP
Solution:
Explanation of Solution
a)
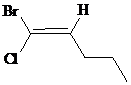
The IUPAC name with
Assign the priority to four groups, –Br gets first priority, pentyl group gets second, –Cl gets third, and –H gets fourth. Since both the priority groups are diagonal to each other, it is
Select the longest carbon chain. The parent hydrocarbon chain is pentane and hence pent- will be used and double bond is at the second position. Therefore, pent-1-ene is used.
Chloro and bromo groups are at first position. So, 1-bromo-1-chloropent-1-ene will be used.
Hence, the IUPAC name of the compound will be
b)
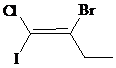
The IUPAC name with
Assign the priority to four groups, –I gets first priority, –Br gets second, –Cl gets third, and ethyl group gets fourth. Since both the priority groups are diagonal to each other, it is
Select the longest carbon chain. The parent hydrocarbon chain is butyl and hence but- will be used and double bond is at the first position. Therefore, but-1-ene is used.
Chloro and iodo groups are at first position and bromo group is at second position. So, 2-bromo-1-chloro-1-iodobut-1-ene will be used.
Hence, the IUPAC name of the compound will be
c)
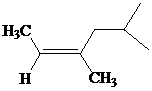
The IUPAC name with
Assign the priority to four groups, 2-methylpropyl gets first priority, both methyl groups get second, and –H gets third. Since both the priority groups are on the same side, it is
Select the longest carbon chain. The parent hydrocarbon chain is hexyl and hence hex- will be used and double bond is at the second position. Therefore, hex-2-ene is used.
There are two methyl groups at third and fifth positions. So, 3,5-dimethylhex-2-ene will be used.
Hence, the IUPAC name of the compound will be
d)
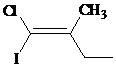
The IUPAC name with
Assign the priority to four groups, –I gets first priority, –Cl gets second, ethyl group gets third, and methyl group gets fourth. Since both the priority groups are on the same side, it is
Select the longest carbon chain. The parent hydrocarbon chain is butyl and hence but- will be used and double bond is at the first position. Therefore, but-1-ene is used.
There is one methyl group at second position, and chloro and iodo groups at first position. So, 1-chloro-1-iodo-2 methylbut-1-ene will be used.
Hence, the IUPAC name of the compound will be
e)
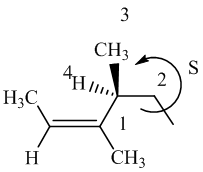
The IUPAC name with
Assign the priority to four groups, 1-methylpropyl gets first priority, both methyl groups get second, and –H gets third. Since both the priority groups are on the same side, it is
Select the longest carbon chain. The parent hydrocarbon chain is hexyl and hence hex- will be used and double bond is at the second position. Therefore, hex-2-ene is used.
There are two methyl groups at third and fourth position. So, 3,4-dimethylhex-2-ene will be used.
Carbon-4 is a chiral carbon. Assigning priority to the groups, 2-butene gets first priority, ethyl gets second, methyl gets third, and hydrogen gets fourth.
Move 1-2-3, it is in anticlockwise direction. So, it is
Hence, the IUPAC name of the compound will be
f)
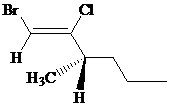
The IUPAC name can be done using the following steps:
Assign the priority to four groups, –Br gets first priority, –Cl gets second, 1-methylbutyl gets third, and –H gets fourth. Since both the priority groups are on the same side, it is
Select the longest carbon chain. The parent hydrocarbon chain is hexyl and hence hex- will be used and double bond is at the first position. Therefore, hex-1-ene is used.
There is one methyl group at third position, and chloro at second and bromo at first position. So, 1-bromo-2-chloro-3-methylhex-1-ene will be used.
Carbon-3 is a chiral carbon. Assigning priority to the groups, 1-bromo-2-chloroethene gets first priority, propyl gets second, methyl gets third, and hydrogen gets fourth.
Rotate the molecule such that –H is at a horizontal position and then move 1-2-3, it is in anticlockwise direction. So, it is
Hence, the IUPAC name of the compound will be
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry
- Compound A and compound B are in equilibrium. Write a stepwise mechanism from compound Ato compound B showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and products are formed. Show all lone pairs and formal charges. Lastly, explain which compound (Aor B) will be in higher concentration.arrow_forwardIn the following three compounds(1,2,3) arrange their relative reactivity towards the reagent CH3Cl / AlCl3. and justify it.arrow_forwardUsing the information provided below, deduce the identity of the compound I, II, III, IV and V. What is the IUPAC name of compound I, II, III, IV and V?arrow_forward
- Compound D and compound E are in equilibrium. Write a stepwise mechanism from compound D to compound E showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and product are formed. Show necessary lone pairs and all formal charges. Lastly explain which compound (D or E) will be in higher concentration.arrow_forwardWhen the nitrogen-containing aromatic heterocyclic compounds 1 and 2 are treated with HCl, only 1 forms the hydrochloride salt, whereas compound 2 is unreactive. Provide an explanation for this observed reactivity.arrow_forwardA) Considering compounds 2a through 2l, identify: 1)one pair of geometric isomers 2)two pairs of enantiomers and 3)three pairs of identical molecules B) Give the names, including the configurations, of each of the geometric isomers and of each of the enantiomers identified in 1A and 1B. Draw the relevant structures. C) Sort compounds 2a, 2b, 2c, 2f and 2k in order of increasing solubility in water and briefly justify.arrow_forward
- Compound A, C3H7Br, does not react with cold dilute potassium permanganate solution. Upon treatment with potassium hydroxide in ethanol, A gives only product B, C3H6. Unlike A, B decolourises potassium permanganate solution. Ozonolysis of Bgives C, C2H4O, and D, CH2O. Suggest the structural formulae of A, B, C and D.Write the equations for all the reactions involved.arrow_forwardgive the structural formula for compounds A to Garrow_forwardA difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired configuration. a. Label this stereogenic center as R or S. b. A well-known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship. d. Suggest a reagent to convert A to the single stereoisomer X.arrow_forward
- Provide a mechanism which explains the following conversions. Include all intermediates (where appropriate) and watch your arrows and chargesarrow_forwardTriphenylmethanol is insoluble in water, but when it is treated with concentrated sulfuric acid, a bright yellow solutionresults. As this yellow solution is diluted with water, its color disappears and a precipitate of triphenylmethanol reappears.Suggest a structure for the bright yellow species, and explain this unusual behavior.1arrow_forwardA difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired conguration.a. Label this stereogenic center as R or S.b. A well known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship.c. Suggest a reagent to convert A to the single stereoisomer X.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
