
Interpretation:
The compound with the molecular formula
Concept introduction:
>The
In dehydrohalogenation
The elimination of primary alkyl halides proceeds through
In
According to Zaitsev rule, the most substituted alkene is the major product.
Answer to Problem 43P
Solution:
The compounds of molecular formula
a)
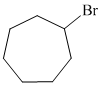
b)
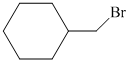
c)
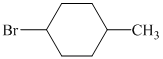
d)
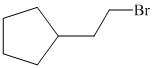
e)
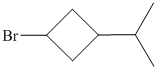
f)
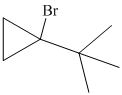
Explanation of Solution
a) The structure of the given alkene as the product of
In
Hence, the structure of alkyl bromide is as follows:

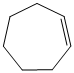
b) The structure of the given alkene as the product of
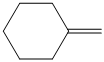
In
So, there are two possible structures of alkyl bromide which are as follows:
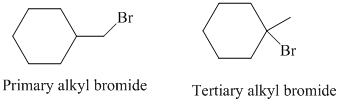
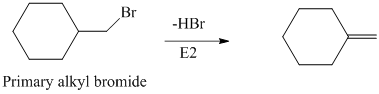
The primary alkyl bromide forms the desired product but the tertiary alkyl bromide can form two alkenes with trisubstituted alkene as the major product. The reaction is as follows:
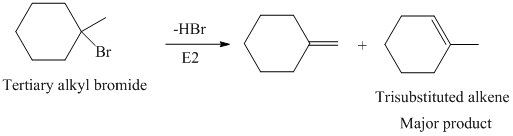
Hence, the structure of the compound that forms the given alkene as an exclusive product of
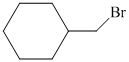
c)The structure of given alkene as the product of
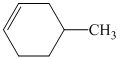
In
So, there are two possible structures of alkyl bromide as follows:
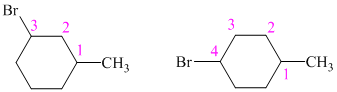
The alkyl bromide in which
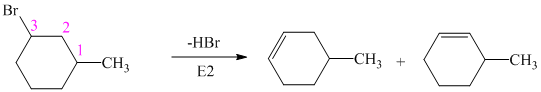
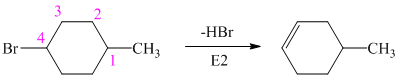
Hence, the structure of the compound that forms the given alkene as an exclusive product of

d) The structure of the given alkene as the product of
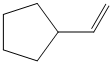
In
So, there are two possible structures of alkyl bromide as follows:
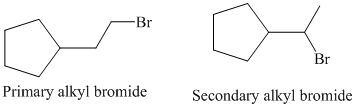
The primary alkyl bromide forms the desired product but the secondary alkyl bromide can form two alkenes with trisubstituted alkene as the major product.
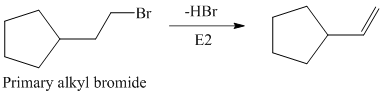
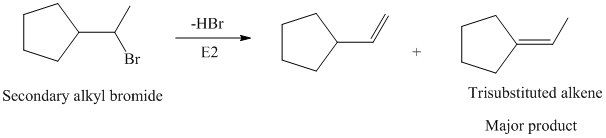
Hence, the structure of the compound that forms the given alkene as an exclusive product of

e) The structural formula of the given alkene is shown below:
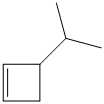
In
So, there are two possible structures of alkyl bromide as follows:
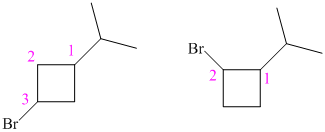
The alkyl bromide in which
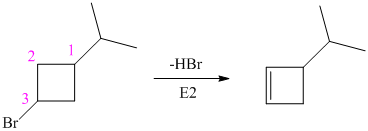
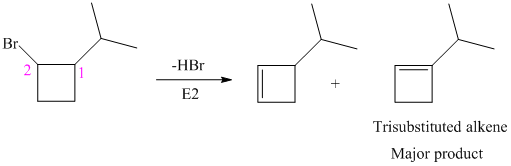
Hence, the structure of the compound that forms the given alkene as an exclusive product of

f) The structural formula of the given alkene is shown below:
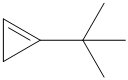
In
So, there are two possible structures of alkyl bromide as follows:
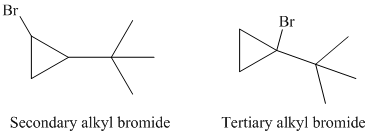
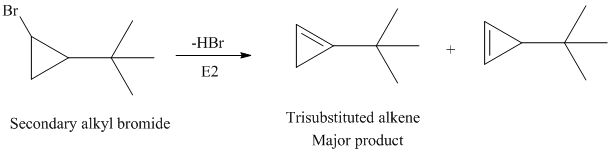
The secondary alkyl bromide forms two alkenesbut the tertiary alkyl bromide forms only the desired product as shown in following equations:
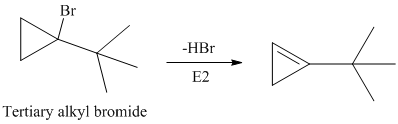
Hence, the structure of the compound that forms the given alkene as an exclusive product of

Want to see more full solutions like this?
Chapter 7 Solutions
ORGANIC CHEMISTRY - CONNECT ACCESS CARD
- Consider the E2 elimination of Compound A, and answer the following questions: (a) Label each stereocenter in Compound A with the correct R or S configuration. (b) Draw the structures of the major product with correct stereochemistry. Assume D and H have the same reactivity.arrow_forwardDehydration of 1,2,2-trimethylcyclohexanol with H2SO4 affords 1-tert-butylcyclopentene as a minor product. (a) Draw a stepwise mechanism that shows how this alkene is formed. (b) Draw other alkenes formed in this dehydration. At least one must contain a ve-membered ring.arrow_forwardSuzuki coupling of aryl iodide A and vinylborane B affords compound C,which is converted to D in the presence of aqueous acid. Identifycompounds C and D and draw a stepwise mechanism for the conversionof C to D.arrow_forward
- An alkene G (C6H12) reacts with cold basic KMnO4 to produce H (C6H14O2). Hydrogenation of Gproduces I (C6H14). Ozonolysis of G produces propanone (CH3COCH3) and propanal(CH3CH2CHO).a) Draw the structure of G, H, I.b) State the reagent(s) and condition for ozonolysis reactionarrow_forwardProvide the missing starting material, reactant or product. Show appropriate stereochemistry. a) b)arrow_forwarda) Draw both chair conformations of the starting alkyl bromide. Indicate which conformation, if either is more stable. Briefly justify your answer. b) The starting alkyl halide has two chiral carbons. What are the configurations (R/S) at each of these carbons? c) Identify the mechanism (E1/E2) which reactions 1, 2, and 3 proceed by. d) Provide mechanisms that explain the products formed in reactions 1, 2, and 3. All electron movement must be shown in order to receive full credit. e) Indicate which product is the major and which product is the minor in reactions 2 and 3. Briefly justify your answer.arrow_forward
- For the following molecule: A. Draw both chair conformations and label substitutents as axial or equatorial B. Indicate which conformation is more stable using equilibrium arrows C. Predict the major product of the reaction with sodium methoxide D. Predict the major product of the reaction with potassium tert-butoxidearrow_forwardWhat alkene yields A and B when it is treated with Br2 in CCl4?arrow_forwardTreatment of compound A (C8H17Br) with NaOCH2CH3 affords two constitutional isomers B and C. Ozonolysis of B affords CH2=O and (CH3CH2CH2)2C=O. Ozonolysis of C affords CH3CH2CH2COCH3 and CH3CH2CHO. What is the structure of A?arrow_forward
- Addition of HCl to alkene X forms two alkyl halides Y and Z. (A) Label Y and Z as a kinetic or thermodynamic product and explain why. (B) Explain why the addition of HCl occurs at the exocyclic C=C, rather than the other C=Carrow_forwardFor alkenes A, B, C, and D: (a) Rank A—D in order of increasing heat of hydrogenation; (b) rank A—D in order of increasing rate of reaction with H2, Pd-C; (c) draw the products formed when each alkene is treated with ozone, followed by Zn, H2O.arrow_forwarda.What product(s) (excluding stereoisomers) are formed when Y is heated with Cl2? b.What product(s) (excluding stereoisomers) are formed when Y is heated with Br2? c.What steps are needed to convert Y to the alkene Z?arrow_forward
