
(a) Using reactions studied in this chapter, show steps by which this
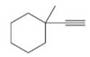
(b) Could the homologous products obtained in these two cases be relied upon to show infrared absorption in the
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
ORGANIC CHEMISTRY-WILEYPLUS ACCESS PKG.
Additional Science Textbook Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Introductory Chemistry (5th Edition) (Standalone Book)
General Chemistry: Atoms First
Living By Chemistry: First Edition Textbook
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry & Chemical Reactivity
- (a) The Friedel-Crafts reaction of benzene with 2-chloro-3-methylbutane in the presence of AlCl3 occurs with a carbocation rearrangement. Give mechanistic explanation and the product formed. (b) Predict the product(s) will be formed from the following reactions: (i) Bromination of p-methylbenzoic acid (ii) Sulphonation of m-bromoanisole (iii) Friedel-craft acylation of o-bromonitrobenzenearrow_forwardFuran undergoes electrophilic aromatic substitution more readily than benzene; mild reagents and conditions are sufficient.For example, furan reacts with bromine to give 2-bromofuran.(a) Propose mechanisms for the bromination of furan at the 2-position and at the 3-position. Draw the resonance forms ofeach sigma complex, and compare their stabilities.(b) Explain why furan undergoes bromination (and other electrophilic aromatic substitutions) primarily at the 2-position.O Br O123furan 2-bromofuranarrow_forward(a) (i) 3-Methyl-2-butanol will react with sulphuric acid to give two isomeric alkenes in 3:1 proportions.(i) Write down the structures for these alkenes and assign appropriate systematic names to them. (ii) Name the most abundant isomer. (iii) Write down detailed mechanism for the formation of a minor product.arrow_forward
- (3) HI does not undergo free radical addition with 1-butene, even in the presence of a peroxide. Please provide an explanation by analyzing the propagation steps if the same radical reaction occurred with HI. Besides the bond dissociation enthalpies in the appendix, the following approximate bond dissociate enthalpies might be useful to you: ~310 kJ/mol The component of C=C CH3CH₂CH₂CH₂-Br CH3CH₂CH₂CH2-I -290 kJ/mol (C-Br bond) ~235 kJ/mol (C-I bond)arrow_forwardGive the reaction mechanism showing all possible products for the following reactions: (a) Reaction of 1-propene with hydrogen bromide, HBr (9) (b) Reaction of ethene with bromine, Br2 (1)arrow_forwardAcid-catalyzed dehydration of neopentyl alcohol, (CH3)3CCH2OH, yields 2-methyl-2- butene as the major product. Outline a mechanism showing all steps in its formation.arrow_forward
- (a) Account for the following :(i) Electrophilic substitution reactions in haloarenes occur slowly.(ii) Haloalkanes, though polar, are insoluble in water.(b) Arrange the following compounds in increasing order of reactivity towards SN2 displacement:2-Bromo-2-Methylbutane, 1-Bromopentane, 2-Bromopentanearrow_forward(b) Answer BOTH parts (i) and (ii). (i) Using your knowledge of silicon chemistry, explain why the synthesis of a compound containing a formal Si=0 double bond is challenging. (ii)Bearing in mind your answer to part (i), suggest a synthetic route to a silanone, R2Si=O, starting from silicon tetrachloride SiCl4 and using any other reagents you choose. Identify all reagents and side-products in your synthetic route and specify your chosen R group, giving reasons for your choice.arrow_forwardThree compounds, A, B, and C, have the same molecular formula, C5H6. In the presence of a platinum catalyst, all three compounds absorb 3 molar equivalents of hydrogen and yield pentane. Compounds B and C give a precipitate when treated with ammoniacal silver nitrate; compound A give no reaction. Compounds A and B show an absorption maximum near 250 nm. Compound C shows no absorption maximum beyond 200 nm. Propose a structure for A, B, and C.arrow_forward
- On being heated with a solution of sodium ethoxide in ethanol, compound A (C7H15Br) produced a mixture of two alkenes B and C, each of which had the molecular formula C7H14. Catalytic hydrogenation of major isomer B or minor isomer C gave only 3-ethylpentane. Suggest structures and mechanisms for compounds A, B, and C consistent with these observations.arrow_forwardThere are several isomeric alkanes of molecular formula C6H14.Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.84 (d, 12 H), 1.39 (septet, 2H) ppm Isomer B: δ = 0.84 (t, 3 H), 0.86 (s, 9H), 1.22 (q, 2H) ppmarrow_forward(a) Write the products of following reactions. Give mechanism with proper stereochemistry? (2 x 2.5 = 5) (ii) Me Δ Δ CNarrow_forward
