
7-10 The rate of disappearance of HCI was measured for the following reaction:
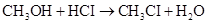
The initial concentration of HCI is 1.85 M. Its concentration decreases to 1.58 M in 54.0 min. What is the
Interpretation:
The rate of given reaction with initial concentration of
Concept introduction:
Rate of reaction: The rate of the reaction is defined as extent of a reaction. For any reaction, it depends on the concentration change of limiting reagent formed in a reaction.
Concentration: Concentration of all chemical species takes part in a reaction depend on their moles present in per liter solution.
The rate is change in concentration of the reactant with change in time. It is mathematically represented as follows:
Answer to Problem 7.10P
Rate of reaction = 0.005 M/min
Explanation of Solution
Given information:
Reaction with initial concentration of
Put the given values in above expression.
Here, negative sign indicates the concentration of HCl decreases.
Rate of reaction = -0.005 M/min
Want to see more full solutions like this?
Chapter 7 Solutions
Bundle: Introduction To General, Organic And Biochemistry, 11th + Owlv2, 1 Term (6 Months) Printed Access Card
- 7-11 Consider the following reaction: Suppose we start the reaction with an initial iodomethane concentration of 0.260 M. This concentration increases to 0.840 M over a period of 1 h 20 min. What is the rate of reaction?arrow_forward7-35 A reaction has a high rate constant but a small equilibrium constant. What does this mean in terms of producing an industrial product?arrow_forward7-13 Why are reactions between ions in aqueous solution generally much faster than reactions between covalent molecules?arrow_forward
- The label on a bottle of 3% (by volume) hydrogen peroxide, H2O2, purchased at a grocery store, states that the solution should be stored in a cool, dark place. H2O2decomposes slowly over time, and the rate of decomposition increases with an increase in temperature and in the presence of light. However, the rate of decomposition increases dramatically if a small amount of powdered MnO- is added to the solution. The decomposition products are H2O and O2. MnO2 is not consumed in the reaction. Write the equation for the decomposition of H2O2. What role does MnO2 play? In the chemistry lab, a student substituted a chunk of MnO2 for the powdered compound. The reaction rate was not appreciably increased. WTiat is one possible explanation for this observation? Is MnO2 part of the stoichiometry of the decomposition of H2O2?arrow_forward7-15 A certain reaction is exothermic by 9 kcal/mol and has an activation energy of 14 kcal/mol. Draw an energy diagram for this reaction and label the transition state.arrow_forwardGo to the PhET Reactions and change to Angled shot to see the difference. (a) What happens when the angle of the collision is changed? (b) Explain how this is relevant to rate of reaction.arrow_forward
- 7-12 Two kinds of gas molecules are reacted at a set temperature. The gases are blown into the reaction vessel from two tubes. In setup A, the two tubes are aligned parallel to each other; in setup B, they are 900 to each other; and in setup C, they are aligned directly opposite each other. Which setup would yield the most effective collisions?arrow_forwardThe reaction of nitrogen dioxide and carbon monoxide in the gas state to form carbon dioxide and nitric oxide is represented by the equation: NO2(g)+CO(g)--->CO2(g)+NO(g) The following data were collected for the above reaction Experiment No [CO2] (M) [CO] (M) Initial Rate (M/s) 1 5.0*10-4 1.6*10-2 1.7*10-7 2 5.0*10-4 3.2*10-2 1.7*10-7 3 1.5*10-3 3.2*10-2 1.5*10-6 Determine the order of the reaction with respect to each reactant.arrow_forwardThe graph below shows a plot of the rate of a reaction versus the concentration of the reactant (Figure 1). c) Estimate the value of k.arrow_forward
- What is the rate of reaction for the chemical reaction, 2H2O2(aq)➡️ 2H2O(I) + O2(g) Reaction started with 1.9 M H2O2 and ended with 0.5 M after 1.2 minutes.arrow_forwardCalculate the reaction rate for selenious acid, H2SeO3, if 0.1150 M I-1 decreases to 0.0770 M in 12.0 minutes. H2SeO3(aq) + 6I-1(aq) + 4H+1(aq) ⟶Se(s) + 2I3-1(aq) + 3H2O(l) Group of answer choices 0.00317 M/min 0.00633 M/min 0.00159 M/min 0.0190 M/min 0.000528 M/minarrow_forwardwhat is the reaction order for A --> B + C if rate = 1 x 10^-5 when [A] = 0.1 M and rate = 9 x 10^-5 when [A] = 0.3Marrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning


