
Concept explainers
(a)
Interpretation: The name of the given compound has to be stated.
Concept introduction: In a cyclic monosaccharide, the replacement of an
(a)
Answer to Problem 7.110EP
The name of the given structure is ethyl-
Explanation of Solution
The Fisher projection formula for D-allose is,
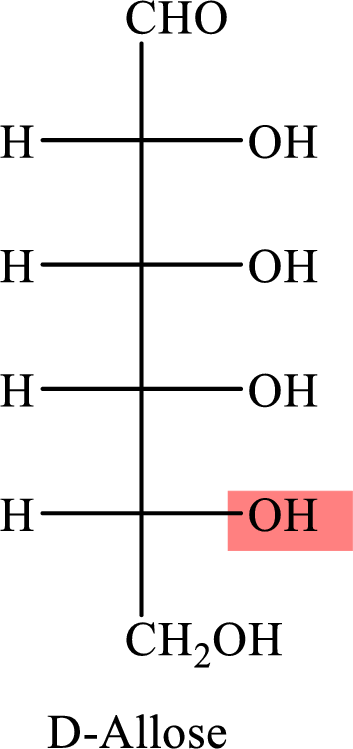
The structure given in Problem 18-104 is,
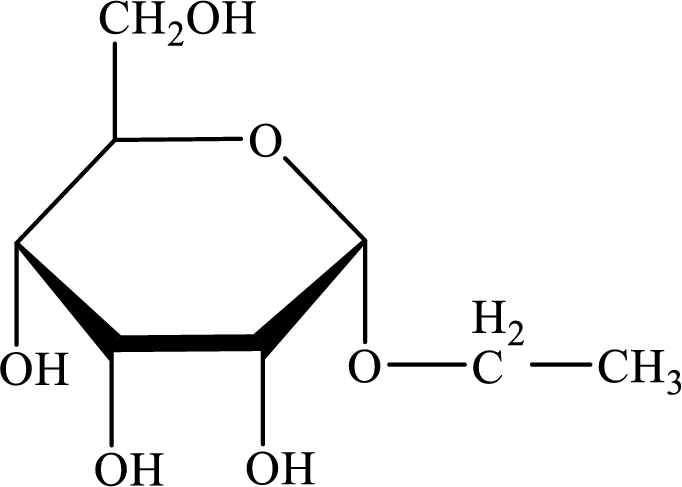
The above structure is the cyclic form of monosaccharide D-allose. Both the
(b)
Interpretation: The name of the given compound has to be stated.
Concept introduction: In a cyclic monosaccharide, the replacement of an
(b)
Answer to Problem 7.110EP
The name of the given structure is methyl-
Explanation of Solution
The Fisher projection formula for D-allose is,
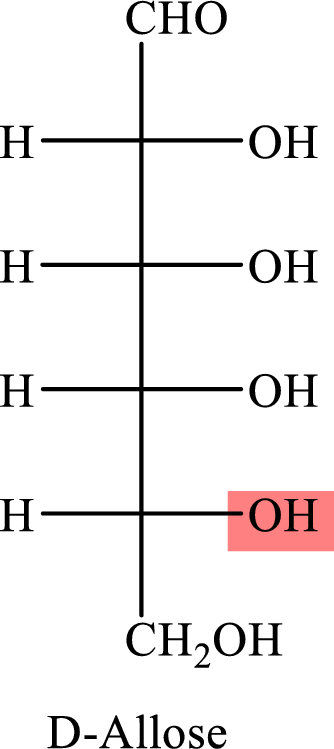
The structure given in Problem 18-104 is,
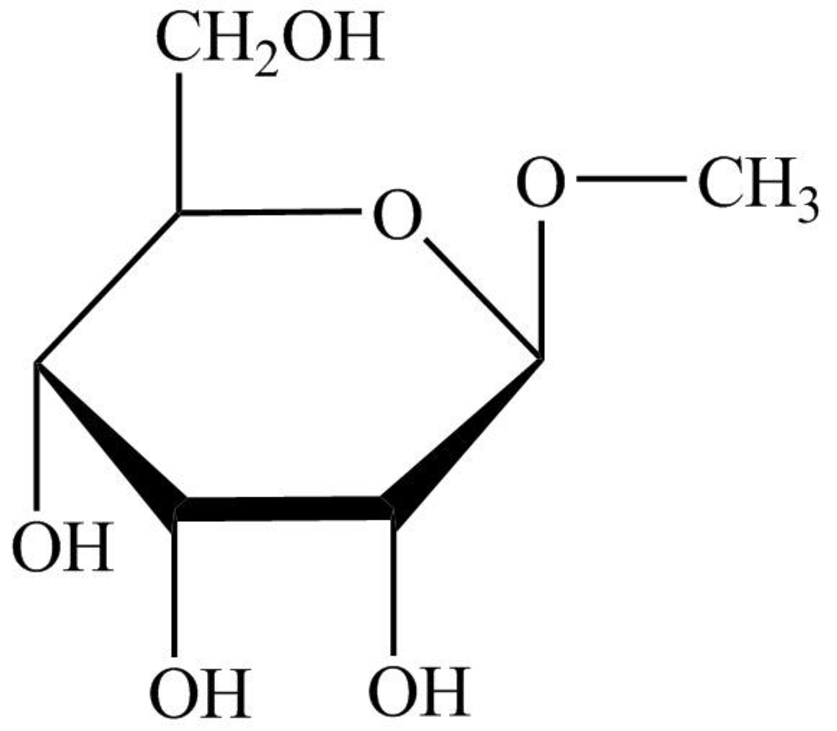
The above structure is the cyclic form of monosaccharide D-allose. Both the
(c)
Interpretation: The name of the given compound has to be stated.
Concept introduction: In a cyclic monosaccharide, the replacement of an
(c)
Answer to Problem 7.110EP
The name of the given structure is ethyl-
Explanation of Solution
The Fisher projection formula for D-psicose is,
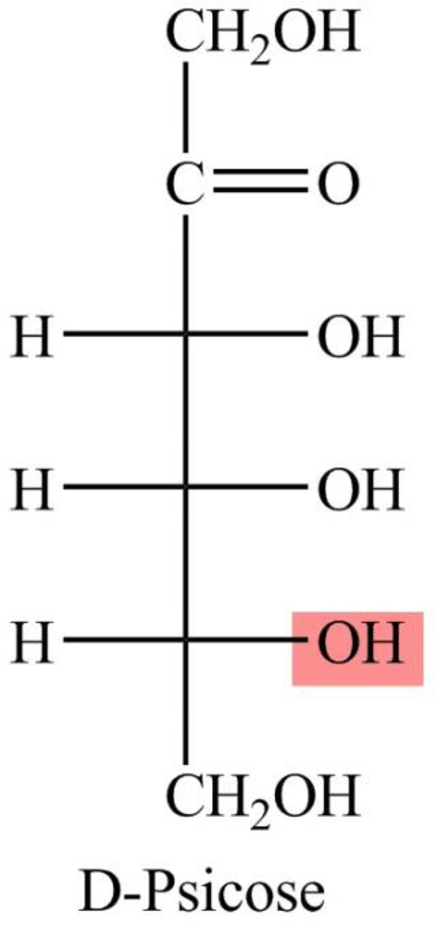
The structure given in Problem 18-104 is,
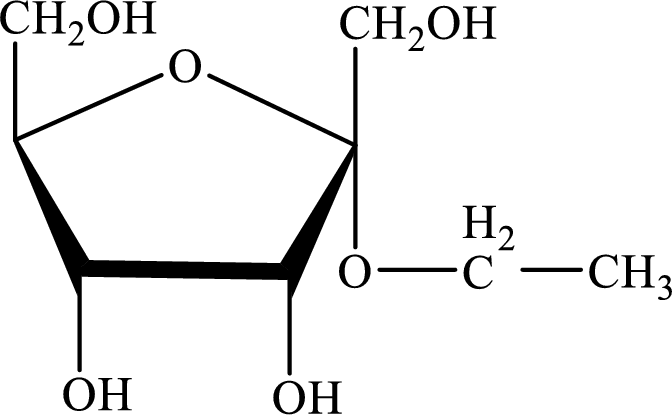
The above structure is the cyclic form of monosaccharide D-psicose. Both the
(d)
Interpretation: The name of the given compound has to be stated.
Concept introduction: In a cyclic monosaccharide, the replacement of an
(d)
Answer to Problem 7.110EP
The name of the given structure is methyl-
Explanation of Solution
The Fisher projection formula for D-mannose is,
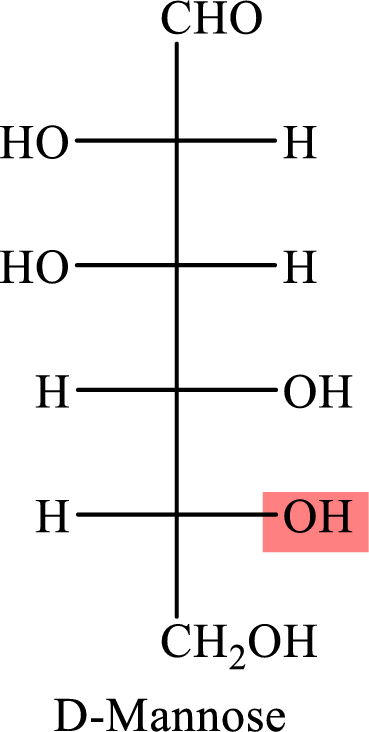
The structure given in Problem 18-104 is,
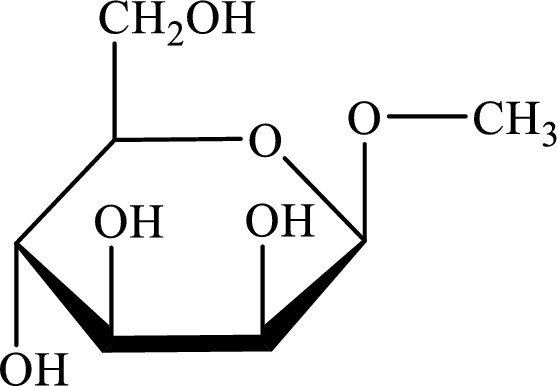
The above structure is the cyclic form of monosaccharide D-mannose. Both the
Want to see more full solutions like this?
Chapter 7 Solutions
EBK ORGANIC AND BIOLOGICAL CHEMISTRY
- Name each of the compounds in Problem 15-106 in the manner described in Section 15-11.arrow_forward1 Hyaluronic acid acts as a lubricant in the synovial fluid of joints. In rheumatoid arthritis, inflammation breaks hyaluronic acid down to smaller molecules. Under these conditions, what happens to the lubricating power of the synovial fluid?arrow_forwardWhich of the following statements is not true for vital force theory? O Life force was necessary to produce organic compounds Organic compounds can only be produced by and within living things O Inorganic materials did not contain the "vital force" of life None of the abovearrow_forward
- (b) Glucose has the following structure. Describe the properties based on the structure of glucose.arrow_forwardComplete the equation, name the initial compounds. What class of organic compounds does the product belong to? O CH₂OH/H+ Harrow_forwardName the organic reactant required to react with ammonia to produce hexan-1-amine and HBr.arrow_forward
- Indicate whether or not each of the following statements is TRUE or FALSE. Justify your answer with ONE or TWO sentences. i. Lipids are considered to be more soluble in water than in other solvents. ii. The many -OH groups present in carbohydrates such as glucose make them quite insoluble in water and blood. ii. Carbohydrate structure shows that water molecules are connected to carbon atoms. iv. Hydrocarbons contain carbon, hydrogen and oxygen. A monosaccharide can be hydrolyzed to smaller units. V.arrow_forwardWhat organic functional group does terephthalic acid contain?arrow_forwardIndicate whether or not each of the following statements is TRUE or FALSE. Justify your answer with ONE or TWO sentences. i. Lipids are considered to be more soluble in water than in other solvents. ii. The many -OH groups present in carbohydrates such as glucose make them quite insoluble in water and blood. iii. Carbohydrate structure shows that water molecules are connected to carbon atoms. iv. Hydrocarbons contain carbon, hydrogen and oxygen. v. A monosaccharide can be hydrolyzed to smaller units.arrow_forward
- Which of the following is a fatty acid which is a solid at room temperature? OH H. OH OHarrow_forwardWhich of the following has the highest melting point? The questions below refer to the fatty acids shown here: 1 2 3 4 1 2 3 4 OH OH OH OHarrow_forwardConsider the structure of the amino acid L-alanine. Which functional groups are common to all amino acids? H COOH NH₂ CH3arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





