
(a)
Interpretation:
Reaction mechanism has to be proposed for the given conversion.
(b)
Interpretation:
The regioselectivity of carbon-carbon bond formation in the given conversion has to be accounted.
Concept Introduction:
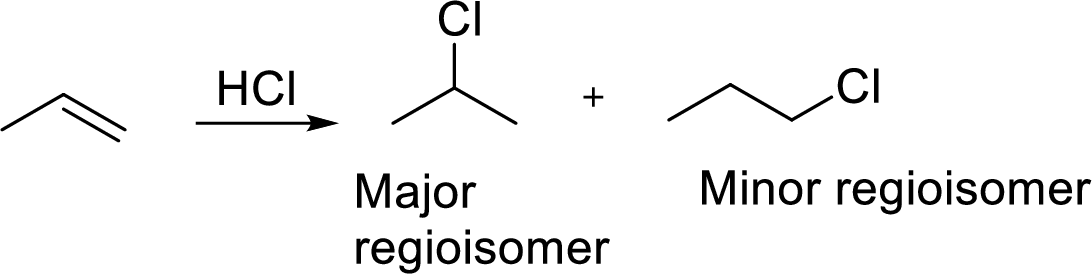
Regioselectivity: It is the favouring of reactants or reagents to bond to one atom or another. Regioisomers are isomers in which connectivity of atoms varies but same number of atoms are present in it. An example of reaction between propene and hydrochloric acid for regioisomers is given as,
(c)
Interpretation:
For the given conversion, experimental condition has to be described.
(d)
Interpretation:
For the given conversion, experimental condition has to be described.
Trending nowThis is a popular solution!

Chapter 7 Solutions
EP ORGANIC CHEMISTRY-OWL V2 ACCESS
- Following is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardPhenylacetone can form two different enols.(a) Show the structures of these enols.(b) Predict which enol will be present in the larger concentration at equilibrium.(c) Propose mechanisms for the formation of the two enols in acid and in basearrow_forwardShow how to synthesize the following amines from the indicated starting materials.(a) N-cyclopentylaniline from anilinearrow_forward
- (D)The best route to synthesise aromatic primary amines is by reduction of the corresponding nitro compounds. Draw the reaction scheme for the preparation of p-toluidine.arrow_forwardShow how to synthesize the following amines from the indicated starting materials.(a) N-cyclopentylaniline from aniline (b) N-ethylpyrrolidine from pyrrolidinearrow_forwardA key step in the hydrolysis of acetamide in aqueous acid proceeds by nucleophilic addition of * OH (a) H3O* to CH3Ĉ NH2 (b) H2O to CH3ČNH2 + OH +OH (c) H3O* to CH,ČNH2 (d) HO¯ to CH3CNH2arrow_forward
- Show how to synthesize the following amines from the indicated starting materials byacylation–reduction.(a) N-butylpiperidine from piperidinearrow_forwardBased on the image attached, it shows methyl salicylate reacts with Ethanamine, and Ether act as a solvent to form N-ethylbenzamide as a product. Explains the mechanism reaction of conversion ester to an amide.arrow_forwardSeveral additional amine syntheses are effectively limited to making primary amines. The reduction of azides and nitrocompounds and the Gabriel synthesis leave the carbon chain unchanged. Formation and reduction of a nitrile adds onecarbon atom. Show how these amine syntheses can be used for the following conversions.(a) allyl bromide S allylamine (b) ethylbenzene S p@ethylanilinearrow_forward
- Propose a reasonable mechanism to explain the transformation of benzene into acetaminophen. EXPLAIN THE MECHANISM Provide textual explanation about reaction Provide clear handwriting and clear imagearrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

