
Introduction To General, Organic, And Biochemistry
12th Edition
ISBN: 9781337571357
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 7.1, Problem 7.1QC
Problem 7-1
In the reaction
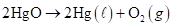
we measure the evolution of oxygen gas to determine the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Exercises20A.1(a) Predict how the total pressure varies during the gas-phase reaction2 ICl(g)+H2(g)→I2(g)+2 HCl(g) in a constant-volume container.20A.1(b) Predict how the total pressure varies during the gas-phase reactionN2(g)+3 H2(g)→2 NH3(g) in a constant-volume container.20A.2(a) The rate of the reaction A+2 B→3 C+D was reported as2.7moldm−3 s−1. State the rates of formation and consumption of theparticipants.20A.2(b) The rate of the reaction A+3 B→C+2 D was reported as2.7moldm−3 s−1. State the rates of formation and consumption of theparticipants.20A.3(a) The rate of formation of C in the reaction 2 A+B→2 C+3 Dis 2.7moldm−3 s−1. State the reaction rate, and the rates of formation orconsumption of A, B, and D.20A.3(b) The rate of consumption of B in the reaction A+3 B→C+2 Dis 2.7moldm−3 s−1. State the reaction rate, and the rates of formation orconsumption of A, C, and D.20A.4(a) The rate law for the reaction in Exercise 20A.2(a) was found to bev=kr[A][B]. What are the units of kr…
pls answer d, e, f thank u
2NO(g)+O2(g)⇌2NO2(g) + Heat
a) where will the reaction be shifted (right or left) if there is an increase in temperature?
b) increase in pressure
c) increase in volume
d.) addition of catalyst
e.) cooling the system
f.) What happens to the concentration of NO2 if there is a decrease in pressure?
g.) removal of NO2
h.) adding more O2
i.) removal of NO?
Determine if each of the following statements is true or false (answer T or F).
a) A change in temperature will change the value of the equilibrium constant for a 'double-arrow' reaction.
b) For an endothermic reaction, the value of Keq (equilibrium constant) will increase if the temperature is increased.
c) The value of the rate constant is negative for a 2nd order decomposition reaction.
d) A buffer can be prepared by mixing equal amounts of a strong acid and a strong base.
e) A catalyst increases the rate of a reaction by lowering the value of the equilibrium constant.
please answer all
Chapter 7 Solutions
Introduction To General, Organic, And Biochemistry
Ch. 7.1 - Problem 7-1 In the reaction we measure the...Ch. 7.4 - Problem 7-2 Calculate the rate for the reaction in...Ch. 7.6 - Prob. 7.3QCCh. 7.6 - Prob. 7.4QCCh. 7.6 - Prob. 7.5QCCh. 7.7 - Prob. 7.6QCCh. 7.7 - Problem 7-7 Consider the following equilibrium...Ch. 7.7 - Prob. 7.8QCCh. 7.7 - Prob. 7.9QCCh. 7 - 7-11 Consider the following reaction: Suppose we...
Ch. 7 - 7-12 Two kinds of gas molecules are reacted at a...Ch. 7 - 7-13 Why are reactions between ions in aqueous...Ch. 7 - Prob. 4PCh. 7 - 7-15 A certain reaction is exothermic by 9...Ch. 7 - 7-16 A quart of milk quickly spoils if left at...Ch. 7 - 7-17 If a certain reaction takes 16 h to go to...Ch. 7 - Prob. 8PCh. 7 - Prob. 9PCh. 7 - Prob. 10PCh. 7 - Prob. 11PCh. 7 - 7-22 If you add a piece of marble, CaCO3 to a 6 M...Ch. 7 - Prob. 13PCh. 7 - Prob. 14PCh. 7 - Prob. 15PCh. 7 - 7-26 Write the chemical equations corresponding to...Ch. 7 - Prob. 17PCh. 7 - 7-28 When the following reaction reached...Ch. 7 - 7-29 The following reaction was allowed to reach...Ch. 7 - Prob. 20PCh. 7 - 7-31 Here are equilibrium constants for several...Ch. 7 - 7-32 A particular reaction has an equilibrium...Ch. 7 - Prob. 23PCh. 7 - Prob. 24PCh. 7 - 7-35 A reaction has a high rate constant but a...Ch. 7 - 7-36 Complete the following table showing the...Ch. 7 - Prob. 27PCh. 7 - Prob. 28PCh. 7 - Prob. 29PCh. 7 - 7-40 Is there any change in conditions that change...Ch. 7 - 7-41 The equilibrium constant at 1127°C for the...Ch. 7 - Prob. 32PCh. 7 - 7-43 (Chemical Connections 7A and 7B) Why is a...Ch. 7 - Prob. 34PCh. 7 - 7-45 (Chemical Connections 7C) A painkiller—for...Ch. 7 - 7-46 (Chemical Connections 7D) What reaction takes...Ch. 7 - Prob. 37PCh. 7 - Prob. 38PCh. 7 - Prob. 39PCh. 7 - 7-50 Draw an energy diagram for an exothermic...Ch. 7 - Prob. 41PCh. 7 - Prob. 42PCh. 7 - Prob. 43PCh. 7 - Prob. 44PCh. 7 - Prob. 45PCh. 7 - Prob. 46PCh. 7 - 7-57 Write the reaction to which the following...Ch. 7 - Prob. 48PCh. 7 - Prob. 49PCh. 7 - Prob. 50PCh. 7 - Prob. 51PCh. 7 - Prob. 52PCh. 7 - Prob. 53PCh. 7 - Prob. 54PCh. 7 - Prob. 55PCh. 7 - Prob. 56PCh. 7 - Prob. 57PCh. 7 - Prob. 58PCh. 7 - 7-69 Pure carbon exists is several forms, two of...Ch. 7 - Prob. 60PCh. 7 - 7-71 You have a beaker that contains solid silver...Ch. 7 - Prob. 62PCh. 7 - Prob. 63PCh. 7 - Prob. 64PCh. 7 - Prob. 65PCh. 7 - Prob. 66PCh. 7 - Prob. 67PCh. 7 - Prob. 68PCh. 7 - Prob. 69PCh. 7 - Prob. 70PCh. 7 - Prob. 71PCh. 7 - Prob. 72PCh. 7 - Prob. 73PCh. 7 - Prob. 74PCh. 7 - 7-82 An equilibrium mixture of O2, SO2, and SO3...Ch. 7 - Prob. 76PCh. 7 - Prob. 77P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Problem 7-2 Calculate the rate for the reaction in Example 7-2 when the initial concentration of H2O2 is 0.36 moI/L.arrow_forwardConsider the following potential-energy curves for two different reactions: a. Which reaction has a higher activation energy for the forward reaction? b. If both reactions were run at the same temperature and have the same orientation requirements to react, which one would have the larger rate constant? c. Are these reactions exothermic or endothermic?arrow_forwardA reaction believed to be either first or second order has a half-life of 20 s at the beginning of the reaction but a half-life of 40 s sometime later. What is the order of the reaction?arrow_forward
- Answer plz fast The balanced equation for this reaction is shown below: 3 NaHCO3(aq) + H3C3H5O7(aq) → Na3C3H5O7(aq) + 3 CO2(g) + 3 H20(l) Devise an experiment to determine the overall order of reaction (be specific in your answer)...arrow_forwardAnswer plz ! The balanced equation for this reaction is shown below: 3 NaHCO3(aq) + H3C3H5O7(aq) → Na3C3H5O7(aq) + 3 CO2(g) + 3 H20(l) Devise an experiment to determine the overall order of reaction (be specific in your answer)?arrow_forwardExperiment Initial [Br–] (M) Initial [BrO3–] (M) Initial [H+] (M) Rate of Disappearance of BrO3– (M s-1) 1 0.00100 0.00500 0.100 2.50x10-4 2 0.00200 0.00500 0.100 5.00x10-4 3 0.00100 0.00750 0.100 3.75x10-4 4 0.00100 0.01500 0.200 3.00x10-3 Determine the order of the reaction for [H+]arrow_forward
- The iodide-ion-catalyzed decomposition of hydrogen peroxide, H2O2, is believed to follow the mechanism H2O2+Ik1H2O+IO(slow)H2O2+IOk2H2O+O2+I(fast) What rate law is predicted by this mechanism? Explain.arrow_forwardConsider the dimerization reaction 2A ---> A_2 rate=k[A]^2 When the initial concentration of A is 2.0 M, it requires 30 minutes for 60% of A to react. Calculate the rate constant at these conditions (CORRECT ANSWER is 4.2 x 10^-4 M/s) please help show stepsarrow_forwardPlease check! 1.Determine the overall reaction order for the reaction, A+B→2C using the table of data below: Experiment Initial [A]/M Initial [B]/M Initial rate/M s-1 1 0.010 0.040 0.0031 2 0.010 0.020 0.0015 3 0.020 0.020 0.0032 a. 0 b. 1 c. 2 <-- I think it's this one b/c the numbers are doubled in the table. d. 3 2. The overall order for the reaction, A+B→C, is 2. A student tests this by measuring the reaction rate at one concentration of A and B, then doubling both concentrations at the same time and re-measuring the rate, which quadrupled. The student concludes that the data support the hypothesis that the above rate law is rate=k[A][B]. Analyze this. a. The data do support the hypothesis but each trial should be repeated. b. The data contradict the hypothesis because the rate should have doubled, not quadrupled. <-- c. The data support the hypothesis but a trial holding one reactant constant is needed. b. The data contradict the hypothesis but testing by…arrow_forward
- 1. For a reaction in which the reactants are gases, which of the following would increase the rate of chemical reaction? (Select all Correct Answers) 2. For a reaction in which one of the reactants is a solid, what would increase the rate of reactionarrow_forwardConsider the decomposition of dinitrogen tetroxide, N2O4, to nitrogen dioxide, NO2: N2O4(g)2NO2(g) How would you expect the spontaneity of the reaction to behave with temperature change?arrow_forwardThe reaction 2 N2O5 (g) → 4 NO2 + O2 (g) has an activation energy of 104 kJ/mol. The value of the rate constant for the reaction is 2.72E-4 s-1 at 40.0 °C.What is the value of the rate constant at 55.0 °C?Answer ___ s-1arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning
Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY