
ORGANIC CHEMISTRY WITH WILEY PLUS; ORGAN
4th Edition
ISBN: 9781119493426
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 7.3, Problem 4ATS
Interpretation Introduction
Interpretation: The Cameroonanol has woody fragrance, and it can be synthesized by deprotonation of compound 1 in the presence of strong base via
mechanism. The curved arrows are to be interpreted for the given conversion:
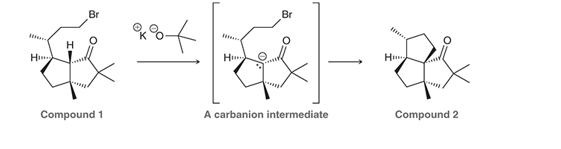
Concept introduction:
In the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Alpha-phellandrene, C₁0H₁6, is a pleasant-smelling chiral compound that may be obtained from the leaves
of eucalyptus trees. It has the molecular formula C₁0H16 and a UV absorption maximum at 263 nm. On
hydrogenation with a palladium catalyst, an achiral hydrocarbon with the formula C₁0H20 is obtained.
Ozonolysis of alpha-phellandrene, followed by treatment with zinc and acetic acid, produces the
following two compounds:
0
||
CH3C-CH
0
||
●
Propose a structure for alpha-phellandrene.
(+
||
HCCH₂CHCH
I
CH(CH3)2
• You do not have to consider stereochemistry.
You do not have to explicitly draw H atoms.
• In cases where there is more than one answer, just draw one.
n [ ]#
?
Compound 1 has been shown to be a useful precursor in the synthesis of natural products. (Org. Lett. 2004, 6, 4439-4442). In
principle, four stereoisomers are possible when this compound is subjected to catalytic hydrogenation. Draw these stereoisomers
and describe their relationships.
HO
O
OH
H₂ - ?
Pt
Modify the four structures below, as necessary, to draw the four stereoisomers that are possible from catalytic hydrogenation of
compound 1. Use the single bond tool to interconvert between double and single bonds.
Treatment of a hydrocarbon A (molecular formula C9H18) with Br2 in the presence of light forms alkyl halides B and C, both having molecular formula C9H17Br. Reaction of either B or C with KOC(CH3)3 forms compound D (C9H16) as the major product. Ozonolysis of D forms cyclohexanone and acetone. Identify the structures of A–D.
Chapter 7 Solutions
ORGANIC CHEMISTRY WITH WILEY PLUS; ORGAN
Ch. 7.2 - Prob. 1CCCh. 7.3 - Prob. 2CCCh. 7.3 - Prob. 1LTSCh. 7.3 - Prob. 3PTSCh. 7.3 - Prob. 4ATSCh. 7.3 - Prob. 2LTSCh. 7.3 - Prob. 5PTSCh. 7.3 - Prob. 6ATSCh. 7.3 - Prob. 7CCCh. 7.4 - Prob. 8CC
Ch. 7.5 - Prob. 9CCCh. 7.6 - Prob. 10CCCh. 7.6 - Prob. 11CCCh. 7.7 - Prob. 12PTSCh. 7.7 - Prob. 13PTSCh. 7.7 - Prob. 14ATSCh. 7.7 - Prob. 4LTSCh. 7.7 - Prob. 16ATSCh. 7.7 - Prob. 17CCCh. 7.7 - Prob. 18CCCh. 7.7 - Prob. 5LTSCh. 7.7 - Prob. 19PTSCh. 7.7 - Prob. 20ATSCh. 7.8 - Prob. 21PTSCh. 7.8 - Prob. 22ATSCh. 7.8 - Prob. 23CCCh. 7.8 - Prob. 24CCCh. 7.8 - Prob. 25CCCh. 7.8 - Prob. 26CCCh. 7.8 - Prob. 27CCCh. 7.9 - Prob. 7LTSCh. 7.9 - Prob. 29ATSCh. 7.9 - Prob. 30ATSCh. 7.9 - Prob. 31ATSCh. 7.10 - Prob. 32CCCh. 7.10 - Prob. 33CCCh. 7.10 - Prob. 34CCCh. 7.11 - Prob. 8LTSCh. 7.11 - Prob. 35PTSCh. 7.11 - Prob. 36PTSCh. 7.11 - Prob. 37ATSCh. 7.11 - Prob. 9LTSCh. 7.11 - Prob. 40PTSCh. 7.11 - Prob. 41ATSCh. 7.12 - Prob. 42CCCh. 7.12 - Prob. 43CCCh. 7.12 - Prob. 44CCCh. 7.12 - Prob. 45CCCh. 7.12 - Prob. 46CCCh. 7 - Prob. 47PPCh. 7 - Prob. 48PPCh. 7 - Prob. 49PPCh. 7 - Prob. 50PPCh. 7 - Prob. 51PPCh. 7 - Prob. 52PPCh. 7 - Prob. 53PPCh. 7 - Prob. 54PPCh. 7 - Prob. 55PPCh. 7 - Prob. 56PPCh. 7 - Prob. 57PPCh. 7 - Prob. 58PPCh. 7 - Prob. 59PPCh. 7 - Prob. 60PPCh. 7 - Prob. 61PPCh. 7 - Prob. 64PPCh. 7 - Indicate whether you would use NaOEt or tBuOK to...Ch. 7 - Prob. 68PPCh. 7 - Draw a plausible mechanism for each of the...Ch. 7 - Prob. 70PPCh. 7 - Prob. 71PPCh. 7 - Prob. 72PPCh. 7 - Prob. 73PPCh. 7 - Prob. 74PPCh. 7 - Prob. 77PPCh. 7 - Prob. 78PPCh. 7 - Prob. 81ASPCh. 7 - Prob. 87ASPCh. 7 - Prob. 90ASPCh. 7 - Prob. 91IPCh. 7 - Prob. 92IPCh. 7 - Prob. 93IPCh. 7 - Prob. 94IPCh. 7 - Prob. 95IPCh. 7 - Prob. 96IPCh. 7 - Prob. 97IPCh. 7 - Prob. 98IPCh. 7 - Prob. 99IPCh. 7 - Prob. 100IPCh. 7 - Prob. 101IPCh. 7 - Prob. 102IPCh. 7 - Prob. 103IPCh. 7 - Prob. 105IPCh. 7 - Prob. 106IPCh. 7 - Prob. 107IPCh. 7 - Prob. 109IPCh. 7 - Prob. 110CPCh. 7 - Prob. 112CPCh. 7 - Prob. 114CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Compounds X has the formula C7H15Cl; Y is C7H15Br.X undergoes base-promoted E2 elimination to give a single alkene product Z. Y likewise reacts under similar conditions to give a single alkene product that is isomeric with ZCatalytic hydrogenation of Z affords 3-ethylpentane.X readily reacts in SN2 fashion with sodium iodide in acetone. Y does not undergo a similar SN2 reaction. Propose structures for X and Y.arrow_forwardThe following reaction was performed as part of a research program sponsored by the National Institutes of Health to develop therapeutic agents for the treatment of cocaine addiction. Using what you have seen about the reactions of halogens with alkenes, propose a mechanism for this process.arrow_forwardIdentify products A and B from the given 1H NMR data. Treatment of acetone [(CH3)2C=O] with dilute aqueous base forms B. Compound B exhibits four singlets in its 1H NMR spectrum at 1.3 (6 H), 2.2 (3 H), 2.5 (2 H), and 3.8 (1H) ppm. What is the structure of B?arrow_forward
- Compound A is first reacted with methylamine in the presence of acid and then treated with NaBH3CN. Using the spectroscopic data given, what is the structure of the product after step 1?arrow_forwardThe reaction of 3,4-dimethyl-3-hexanol (3,4-dimethylhexan-3-ol) with HBr generates compound A as the major product. Treatment of compound A with a strong base gives two isomers of compound B as the major product, along with one isomer of compound C and one isomer of compound D as minor products, all of which have one double-bond equivalent. Identify compounds A, B, C, and D and give their names. By what mechanism does the reaction of 3,4-dimethyl-3-hexanol with HBr occur? By what mechanism does the reaction of A with strong base to form B occur? Propose reaction conditions for an alternative, one-step method for converting 3,4-dimethyl-3-hexanol directly to compound B.arrow_forwardTreatment of cis-4-bromocyclohexanol with HO– affords compound A and cyclohex-3-en-1-ol. Treatment of trans-4- bromocyclohexanol under the same conditions forms compound B and cyclohex-3-en-1-ol. A and B contain different functional groups and are not isomers of each other. Propose structures for A and B and offer an explanation for their formation.arrow_forward
- 3. An alcohol containing natural product A (C10H200) that is a common ingredient in cough drops yields two structurally isomeric alkenes, 1 and 2, on dehydration. Ozonolysis followed by an oxidative workup or a reductive workup as shown yields 3 and 4 from 1 and 2, respectively. Hydroboration of alkene 1 yields the starting alcohol A as the major product from 1. Hydroboration yields a mixture of the starting alcohol A and a diastereomer of A starting from alkene 2. Given this information deduce the structure of A, 1, and 2. Show the correct absolute and relative stereochemistry for A (the alcohol group is cis to a methyl group), 1, and 2 and correctly name these three compounds. H2SO4 heat 1) O3 Н. 1 (C10H18) 2. Zn, H3O* 3 H. A (C10H200) H2SO4 1) O3 н СНО heat 2 (C10H18) 2. Zn, H3O* Н 4arrow_forwardGrignard reagent is a versatile tool in synthetic organic chemistry. Using bromocyclopentane as a starting material, show how a Grignard reagent, X, is synthesized. Reaction of X with water produces compound Y while treatment in carbon dioxide followed by hydrolysis forms compound Z. 3-methyl-2butanone reacts with X and hydrolyses to yield compound AA. Draw the structural formulae of compounds Y, Z and AA and write the chemical equations respectively.arrow_forwardFollowing is the structure of miconazole, the active antifungal agent in a number of over-the-counter preparations, including Monistat, that are used to treat vaginal yeast infections. One of the compounds needed for the synthesis of miconazole is the trichloro derivative of toluene shown on its right. (a) Show how this derivative can be synthesized from toluene. (b) How many stereoisomers are possible for miconazole?arrow_forward
- -Ocimene is a pleasant-smelling hydrocarbon found in the leaves of certain herbs. It has the molecular formula C10H16 and a UV absorption maximum at 232 nm. On hydrogenation with a palladium catalyst, 2,6-dimethyloctane is obtained. Ozonolysis of -ocimene, followed by treatment with zinc and acetic acid, produces the following four fragments: (a) How many double bonds does -ocimene have? (b) Is -ocimene conjugated or nonconjugated? (c) Propose a structure for -ocimene. (d) Write the reactions, showing starting material and products.arrow_forwardA step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forwardThe following bicyclic ketone has two -carbons and three -hydrogens. When this molecule is treated with D2O in the presence of an acid catalyst, only two of the three -hydrogens exchange with deuterium. The -hydrogen at the bridgehead does not exchange. How do you account for the fact that two -hydrogens do exchange but the third does not? You will find it helpful to build models of the enols by which exchange of -hydrogens occurs.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,