
Concept explainers
(a)
Interpretation: The order of stability is to be interpreted for the given
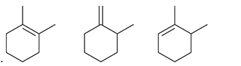
Concept introduction:
(b)
Interpretation: The order of stability is to be interpreted for the given alkenes:
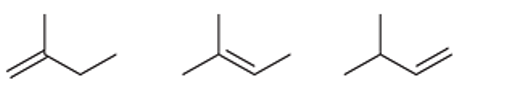
Concept introduction:
Unsaturated hydrocarbons with at least one double bond between the carbon atoms are known as alkenes. The presence of pi bonds in these molecules makes them more reactive compared to saturated hydrocarbons; alkanes. The stability of alkenes depends on their substitution.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
ORGANIC CHEM PRINT STUDY GDE & SSM
- Which one alkene will exhibit cis-trans isomerism? A B OC OA u B Darrow_forwardDehydrohalogenation of 1-chloro-1-methylcyclopropane affords two alkenes (A and B) as products. Explain why A is the major product despite the fact that it contains the less substituted double bond.arrow_forwardFive isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to give alkyl halides having molecular formulaC6H13Cl. A yields five constitutional isomers. B yields four constitutionalisomers. C yields two constitutional isomers. D yields threeconstitutional isomers, two of which possess stereogenic centers. Eyields three constitutional isomers, only one of which possesses astereogenic center. Identify the structures of A–E.arrow_forward
- Draw the alternative chair conformations for the product formed by the addition of bromine to 4-tert-butylcyclohexene. The Gibbs free energy differences between equatorial and axial substituents on a cyclohexane ring are 21 kJ (4.9 kcal)/mol for tert-butyl and 2.0–2.6 kJ (0.48–0.62 kcal)/mol for bromine. Estimate the relative percentages of the alternative chair conformations you drew in the first part of this problemarrow_forwardRank the following alkenes from the most to the least stable.arrow_forward1. Illustrate details mechanism of reactions for bromination of the following alkane. State each of the steps involves. CH3 Br2 CH `CH3 hv, 25°C H3C 2. Draw the two chair conformations of cis-1-chloro-4-methylcyclohexane. Which of the conformers is more stable?arrow_forward
- State what alkene or alkyne (with only carbons and hydrogens) and what other reactants you should use to most effectively synthesize the following in the fewest number of steps and with the highest yield. H;C-CH,- C CH3arrow_forwardG.133.arrow_forwardThe low melting point of oils (like olive oil) is due to what characteristic of its stereochemistry? cis-alkene geometry trans-alkene geometry R configuration OS configurationarrow_forward
- The catalytically active species responsible for the catalytic conversion of the compound * :CH2=CH-CH2OH to CH3-CH=CHOH isarrow_forwardName the alkenes below. Use ONLY E/Z designators to indicate stereochemistry. H3C H Ja H CH₂-C H CH3 CH₂CH3arrow_forwardWhat type of stereochemistry does catalytic hydrogenation afford? Anti-Addition Syn-Addition What is the product of a dissolving metal reduction of an alkyne? An alkyne An alkane OA trans-alkene A cis-alkene Reaction of an alkene with potassium permanganate affords: An anti-diol A syn-diolarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

