
Concept explainers
Interpretation:
Among the given options, which one belongs to changes of state is the final state that of solid has to be chosen.
Concept Introduction:
A change of state is a cycle in that a substance is transformed from one physical state to another physical sate. The physical changes of a substance occurs by either heating or cooling. There are six possible changes of state and they are freezing, melting, evaporation, condensation, sublimation, and deposition. The below mentioned figure will represent each of the physical changes of a substance from starting stage to final stage via intermediate stage.
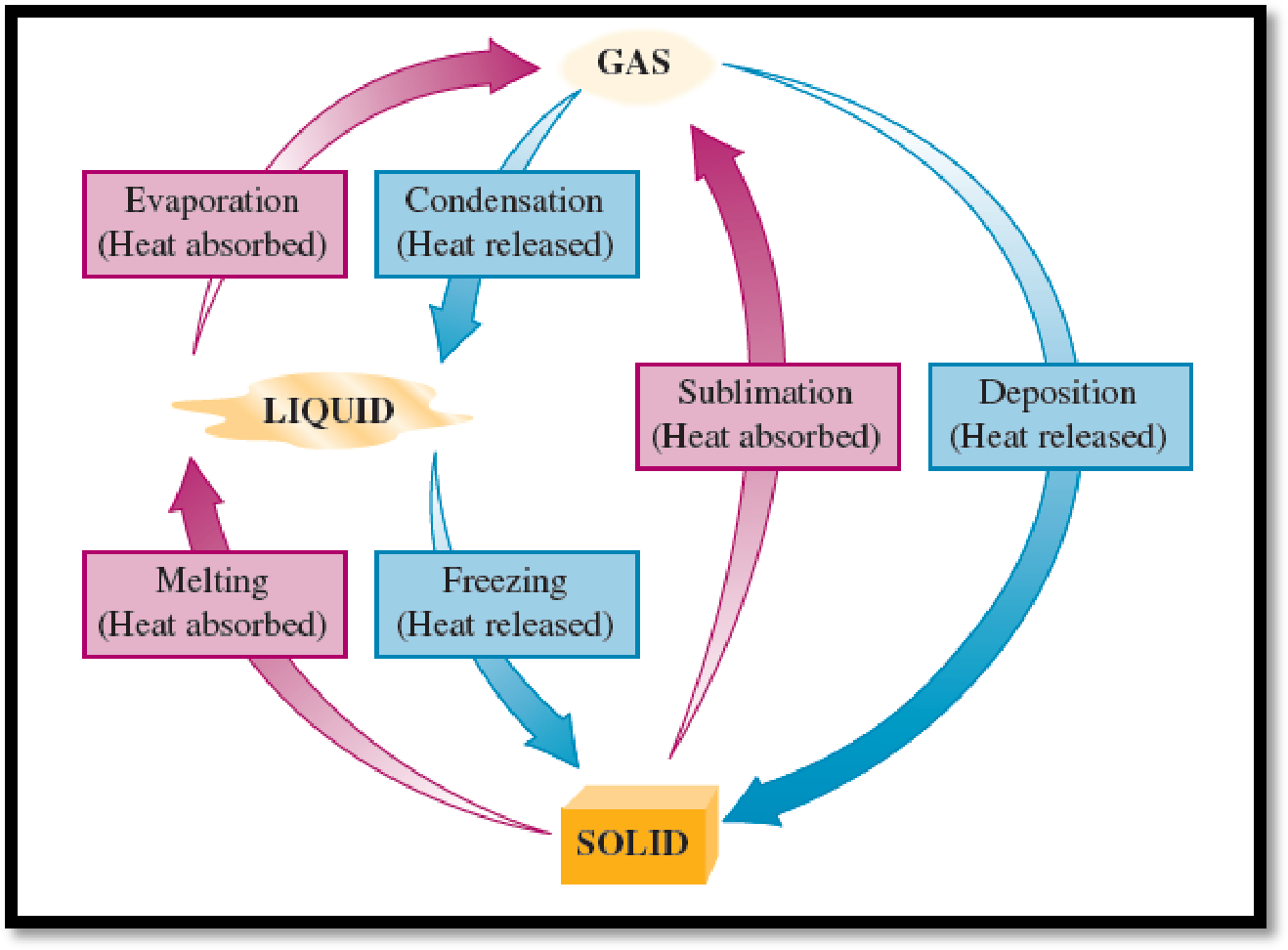
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- trong>Exercise 14.1 Calculate the total energy required to melt 15gof ice at 0C, heat the water to 100C, and vaporize it to steam at 100C.arrow_forwardWhich of the following will occur as a pot of water is heated up on a stove? a. disruptive forces will increase b. molecular speed will increase c. cohesive forces will increase d. more than one response is correctarrow_forwardExercise Determine the degrees of freedom at each indicated point on the phase diagram. (a) Temperature (b) Temperature Pressure Pressurearrow_forward
- Potassium metal has a body-centered cubic structure with all atoms at the lattice points (sec art at left). The density of the metal is 0.856 g/cm3. Calculate the edge length of a unit cell.arrow_forward16. The following surface must remain as a plane and cannot be replaced by another element: * Drive All choices Part Checkarrow_forwardA volatile liquid would A. have weak attractive forces between molecules B. evaporate "slowly" at room temperature C. have a low vapor pressure at room temperature D. more than one correct response E. no correct responsearrow_forward
- Select all covalent network solids Select 2 correct answer(s) ) LiF O C(m) | Si |H2O NaClarrow_forwardTest yourself 6: 1. Name the type of intermolecular forces that exists between the following pairs a) HBr and H,S b) I and NH, TERM ONE• MODULE 2arrow_forwarda. Calculate the heat required to melt 5.87 g of benzene at its normal melting point. Heat of fusion (benzene) = 9.92 kJ/mol Heat = b. Calculate the heat required to vaporize 5.87 g of benzene at its normal boiling point. Heat of vaporization (benzene) = 30.7 kJ/mol Heat = Submit Answer Try Another Version 5 item attempts remainingarrow_forward
- Define boiling point and normal boiling point. The boiling point is the temperature at which the liquid boils and the vapor pressure of the liquid is equal to v the prevailing atmospheric pressure. The normal boiling point is the boiling point of a liquid when the atmospheric pressure is mm Hg. eTextbook and Media Hint Save for Later Attempts: 1 of 15 used Submit Answer MacBook Air DII DD 888 F6 F7 F8 F9 F10 F3 F4 F5 F2 # 2$ & 3 4 7 8arrow_forwardFigure 11.35 shows solid dots (atoms) forming a two-dimensional lattice. A unit cell is marked off by the black line. How many atoms are there in this unit cell in such a two-dimensional lattice? Figure 11.35 A two-dimensional latticearrow_forwardShown here is a representation of a unit cell for a crystal. The orange balls are atom A, and the gray balls are atom B. a What is the chemical formula of the compound that has this unit cell (A? B?)? b Consider the configuration of the A atoms. Is this a cubic unit cell? If so, which type?arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


