
Loose Leaf for Organic Chemistry
10th Edition
ISBN: 9781259626548
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education (edition 10)
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 57P
Dehydration of
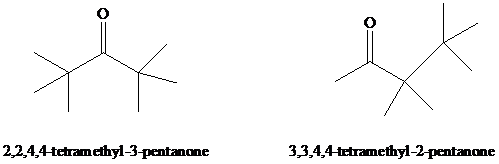
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A. In the synthesis of 1-bromobutane, what is the inorganic by-product left in the reaction flask following the distillation? Why was the bromoalkane the bottom layer in the separatory funnel?
B. Predict the product when 1-methylcyclohexanol reacts with H2SO4 and KBr. Show the mechanism.
Ozonolysis (O3 in CH2Cl2) of compound A under reducing conditions (Zn /acetic acid) gives formaldehyde, 2-butanone, and compound B. Catalytic hydrogenation (H2/Pd) of A gives 2,7-dimethylnonane. What is a possible structure for compound A?
a. 2,7-Dimethyl-2,8-nonadieneb. 2,7-Dimethyl-1,8-nonadienec. 2,7-Dimethyl-1,6-nonadiened. 2,7-Dimethyl-1,7-nonadiene
Reaction of N,N-diethyl-p-diaminobenzene with sodium nitrite and hydrochloric acid at 0°C, followed by treatment with nitrobenzene.
Chapter 8 Solutions
Loose Leaf for Organic Chemistry
Ch. 8.1 - What three alkenes yield 2-methylbutane on...Ch. 8.2 - Prob. 2PCh. 8.2 - Prob. 3PCh. 8.3 - Prob. 4PCh. 8.4 - Prob. 5PCh. 8.4 - Give a structural formula for the carbocation...Ch. 8.5 - Prob. 7PCh. 8.6 - Instead of the three-step process of Mechanism...Ch. 8.6 - The rates of hydration of the two alkenes shown...Ch. 8.6 - Is the electrophilic addition of hydrogen chloride...
Ch. 8.7 - You can calculate the equilibrium constant for the...Ch. 8.7 - Does the presence or absence of a catalyst such as...Ch. 8.7 - The gas phase reaction of ethanol with hydrogen...Ch. 8.8 - Prob. 14PCh. 8.8 - Hydroborationoxidation of -pinene, like its...Ch. 8.10 - Arrange the compounds 2-methyl-1-butene,...Ch. 8.10 - Give the structure of the product formed when each...Ch. 8.11 - Prob. 18PCh. 8.11 - Prob. 19PCh. 8.12 - Prob. 20PCh. 8.12 - Prob. 21PCh. 8.13 - Prob. 22PCh. 8.14 - Prob. 23PCh. 8.14 - Prob. 24PCh. 8 - How many alkenes yield...Ch. 8 - Prob. 26PCh. 8 - Catalytic hydrogenation of...Ch. 8 - Prob. 28PCh. 8 - Prob. 29PCh. 8 - Prob. 30PCh. 8 - Prob. 31PCh. 8 - A single epoxide was isolated in 7984% yield in...Ch. 8 - Prob. 33PCh. 8 - Prob. 34PCh. 8 - On catalytic hydrogenation over a rhodium...Ch. 8 - Prob. 36PCh. 8 - Prob. 37PCh. 8 - Prob. 38PCh. 8 - Prob. 39PCh. 8 - 1-Butene has a higher heat of hydrogenation than...Ch. 8 - Match the following alkenes with the appropriate...Ch. 8 - The heats of reaction were measured for addition...Ch. 8 - Complete the following table by adding + and -...Ch. 8 - Match the heats of hydrogenation (107 kJ/mol,...Ch. 8 - The iodination of ethylene at 25 C is...Ch. 8 - Specify reagents suitable for converting...Ch. 8 - (a) Which primary alcohol of molecular formula...Ch. 8 - Identify compounds A and B in the retrosynthesis...Ch. 8 - Identify compounds A and B in the retrosynthesis...Ch. 8 - Prob. 50PCh. 8 - On being heated with a solution of sodium ethoxide...Ch. 8 - Compound A (C7H15Br) is not a primary alkyl...Ch. 8 - Prob. 53PCh. 8 - Prob. 54PCh. 8 - A mixture of three alkenes (A, B, and C) was...Ch. 8 - Reaction of 3,3-dimethyl-1-butene with hydrogen...Ch. 8 - Dehydration of 2,2,3,4,4-pentamethyl-3-pentanol...Ch. 8 - Prob. 58PCh. 8 - East Indian sandalwood oil contains a hydrocarbon...Ch. 8 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - On the basis of the mechanism of acid-catalyzed...Ch. 8 - As a method for the preparation of alkenes, a...Ch. 8 - Which of the following is the most reasonable...Ch. 8 - Prob. 68PCh. 8 - Oxymercuration Concerns about mercurys toxicity...Ch. 8 - Prob. 70DSPCh. 8 - Prob. 71DSPCh. 8 - Prob. 72DSPCh. 8 - Prob. 73DSPCh. 8 - Oxymercuration Concerns about mercurys toxicity...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forwardReaction of N,N-diethyl-p-diaminobenzene with sodium nitrite and hydrochloric acid at 0°C and subsequent reaction with nitrobenzene.arrow_forwardCompound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr), which reacts violently with D2O to give 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (CH3COCH3) followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2 SO4 gives E (C10 H16), which decolorizes two equivalents of Br2 to give F (C10H16 Br4). E undergoes hydrogenation with excess H2 and a Pt catalyst to give isobutylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughout.arrow_forward
- Reaction of (Z)-methyl-3-hexene with H2O/H2SO4 produces 3-methyl-3-hexanol (75% yield) 1. Write a balance chemical equation for the reaction. 2. Propose a reaction mechanistic explanation for the reaction.arrow_forwardalkylation of benzene with 1-chlorobutane in the presence of AlCl3 gives both butylbenzene and (1-methylpropyl)benzene as products. Propose a route to butylbenzene from benzene that does not also give the (1-methylpropyl)benzene side-product.arrow_forwardCompound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr) which reacts violently with D2O to give 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (Ch3COCH3) followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2SO4 gives E (C10H16), which reacts with 2 equivalents of Br2 to give F (C10H16Br4). E undergoes hydrogenation with excess H2 and Pt catalyst to give 2-methylpropylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughout.arrow_forward
- 1.Treatment of 3-methyl-2-butanol with hydrogen chloride yielded only a trace amount of 2-chloro-3-methylbutane. An isomeric chloride was isolated in 97% yield. Explain why an isomeric compound is formed instead, andsuggest a reasonable structure for this product. 2) Acid-catalyzed hydration of 2-Methyl-1-butene yields two alcohols. The major product does not undergo oxidation, while the minor product will undergo oxidation. Explain why, by showing the structures of the products. 3) The Williamson ether synthesis cannot be used to prepare tert-butyl phenyl ether. Explain why this method cannot be used in this case. 4) Show the structure of the main product when 2,2-dimethyloxirane reacts in methanol. (a) in the presence of an acid, (b) with added sodium methoxide.arrow_forwardWrite complete chemical equation showing reactants, products, and catalysts needed (if any) for the following reaction: d) Reaction of cis-3,3-Dimethyl-4-propylocta-1,5-diene with two mole of HBr e) Reaction of trans-1-Bromo-3-chlorocyclopentane with potassium hydroxide f) Formation of Gilman reagent using isopropyl bromide g) Ozonolysis of 3,3-Dimethyloct-4-ynearrow_forwardWhat product is obtained from the reaction of excess benzene with 1-chloro-2,2-dimethylpropane + AlCl3 ?arrow_forward
- 7. Compound AA, C4H8O, gives a positive result (formation of a silver mirror) when reacted with Tollen’s reagent. Reduction of compound AA with lithium aluminium hydride, LiAlH4 followed by acidified water, H3O+ produces an alcohol, BB. Oxidation of BB with hot acidified potassium dichromate, K2Cr2O7 gives butanoic acid, C3H7COOH. Esterification between butanoic acid and methanol, CH3OH produces compound CC. Dehydration of compound BB using concentrated sulphuric acid, H2SO4 at a temperature of 180°C produces compound DD. Draw the structural formula of compounds AA, BB, CC, and DDarrow_forwardWhen 1-butanol is treated with conc. H2SO4 and heat .A product 'A' is formed from the reaction. Treatment of 'A' with HCl/H2O gives 'B'. Treatment of 'A' with cold KMnO4/OH- gives 'C' Treatment of 'A' with hot KMnO4 /OH- gives 'D' followed by acidification of the mixture to give 'E' 1. identify the compounds represented by A,B,C,D and E.arrow_forward1. (a) Oxidation of 1-butanol with PCC will produce which of the following? 2-chlorobutanal butanone butanoic acid butanal (b) Which synthetic route is the best way to prepare ethyl isopropyl ether? CH3CH2OH + (CH3)2CHOH + H2SO4, 140 oC CH3CH2ONa + (CH3)2CHOH CH3CH2ONa + (CH3)2CHBr (CH3)2CHONa + CH3CH2Br (c) The Williamson ether synthesis involves nucleophilic substitution involving an alkyl halide and an alcohol. (True or False)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY