
Interpretation:
The thermal efficiency of the cycle and the turbine efficiency for the simple power plant shown should be determined.
Concept introduction:
The steam power plant has the turbine operating adiabatically with the given inlet steam pressure and temperature. We used the values shown in the figure to determine the thermal efficiency and turbine efficiency. Turbine efficiency is determined to be the ratio of enthalpy change in the process to the enthalpy change in the reversible process while thermal efficiency is obtained by taking the ratio of energy released by the system to the heat applied to the system.
Answer to Problem 8.1P
The thermal efficiency of the cycle is determined to be 31.1 % and the turbine efficiency is 79 %.
Explanation of Solution
Given information:
The basic cycle for a steam power plant is shown by Fig. 8.1.
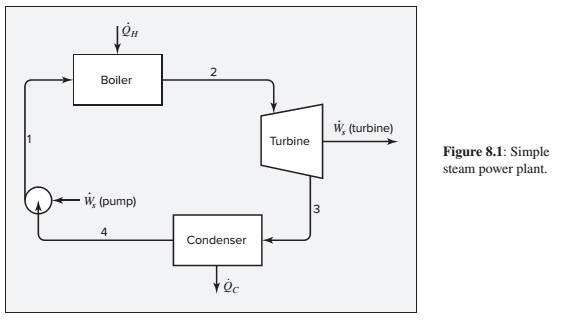
Suppose the turbine operates adiabatically with inlet steam at 6800 kPa and 550°C, and the exhaust steam enters the condenser at 50°C with a quality of 0.96. Saturated liquid water leaves the condenser, and is pumped to the boiler. Neglecting pump work and kinetic- and potential-energy changes, we need to determine the thermal efficiency of the cycle and the turbine efficiency.
Turbine efficiency is determined to be the ratio of enthalpy change in the process to the enthalpy change in the reversible process while thermal efficiency is obtained by taking the ratio of energy released by the system to the heat applied to the system.
Calculation:
Turbine efficiency is given by the formula
Where
Here
We know that
We know that
Enthalpy of steam after neglecting work done is
Therefore, the turbine efficiency is
And the thermal efficiency of the cycle is
The thermal efficiency of the cycle is determined to be 31.1 % and the turbine efficiency is 79 %.
Want to see more full solutions like this?
Chapter 8 Solutions
INTRO.TO CHEM.ENGR.THERMO.-EBOOK>I<
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





