
Concept explainers
(a)
Interpretation:
The molecular orbital diagram of
(a)
Explanation of Solution
The electronic configuration is,
The molecular orbital diagram of
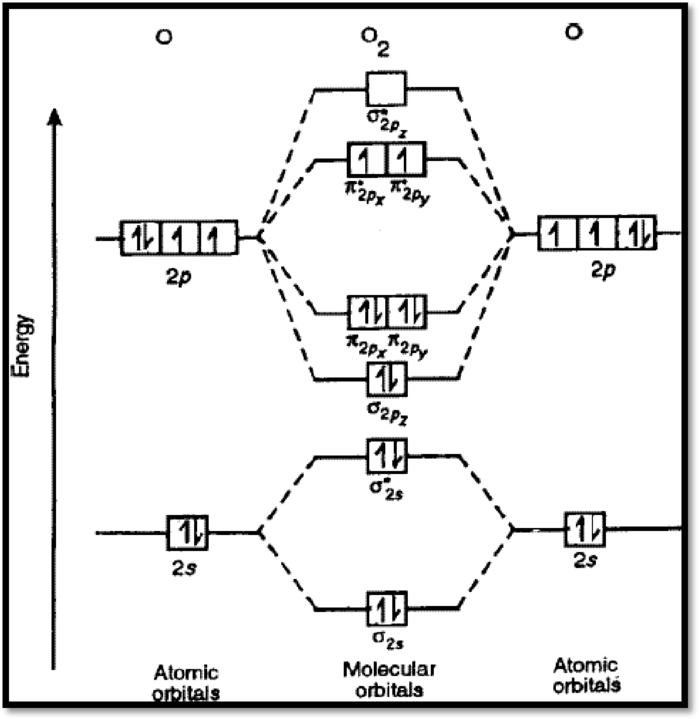
Figure 1
The bond order of
The bond order of
The magnetic nature of
(b)
Interpretation:
The molecular property of oxygen that is explained by its molecular orbital diagram but not by Lewis structure has to be given.
(b)
Explanation of Solution
The molecular property of oxygen that is explained by its molecular orbital diagram is magnetic property.
The Lewis structure of oxygen is,

From the Lewis structure, it can be seen that all electrons are paired, and it is diamagnetic in nature. However, the molecular orbital diagram of oxygen (figure 1) shows the presence of two unpaired electrons and therefore, oxygen is paramagnetic.
(c)
Interpretation:
The nature of highest occupied molecular orbital in oxygen has to be given.
(c)
Explanation of Solution
The electronic configuration of oxygen in the ground state is
(d)
Interpretation:
The bond order and magnetic property in peroxide ion and superoxide ion has to be given.
(d)
Explanation of Solution
The formula of peroxide ion is
The formula of superoxide ion is
Superoxide ion
The molecular orbital diagram of
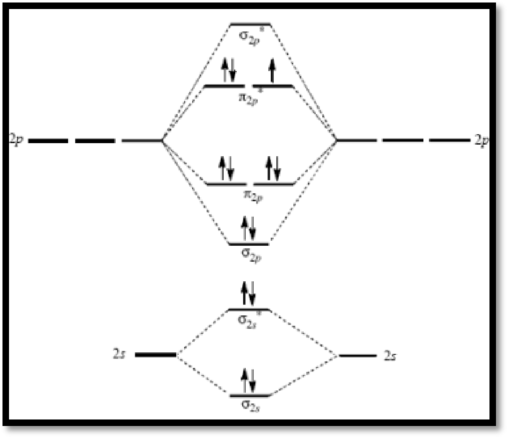
Figure 2
The bond order of
Superoxide ion
Peroxide ion
The molecular orbital diagram of
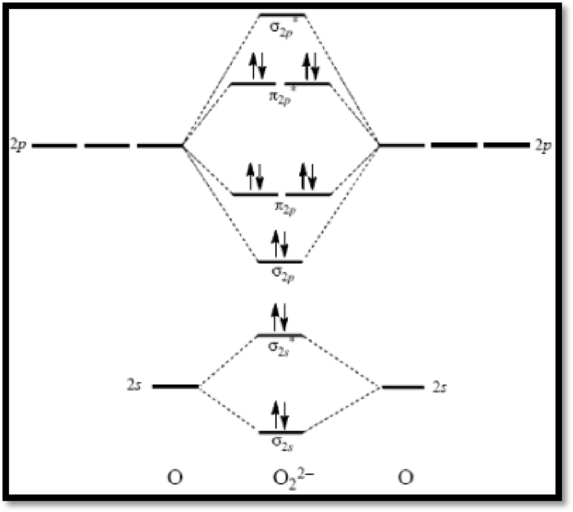
Figure 3
The bond order of
Peroxide ion
Want to see more full solutions like this?
Chapter 8 Solutions
CHEMICAL PRINCIPLES W/SAPLING
- Write two balanced chemical equations in which sulfuric acid acts as an oxidizing agent.arrow_forwardWhich is the stronger acid, H2SO4 or H2SeO4? Why? You may wish to review the Chapter on acid-base equilibria.arrow_forwardDescribe the hybridization and the bonding of a silicon atom in elemental silicon.arrow_forward
- Predict the outcome of the following reactions and explain the reason briefly.arrow_forwardWrite the chemical formula for each of the following compounds, and indicate the oxidation state of the halogen or noble-gas atom in each hydroiodic acidarrow_forwardWrite the balanced reaction for the synthesis of Al2O3 from its elements?arrow_forward
- Which of these two oxides, SO3 and P4O10 , when reacts with water will produce stronger acid?arrow_forwardTreatment of cobalt(II) oxide with oxygen at high temperatures gives Co3O4. Write a balanced chemical equation for this reaction. What is the oxidation state of cobaltin Co3O4?arrow_forwardHow would you expect the structure of the ozonide ion, O3-, to differ from that of ozone?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning



