
Concept explainers
(a)
Interpretation:
The apparatus has to be redrawn if the temperature is increased from
Concept introduction:
Charles’s law: States that volume is directly proportional to temperature when the gas is held at constant pressure and number of molecules.
(a)
Answer to Problem 8.26UKC
According to Charles’s law, if temperature increases at constant pressure, then volume also increases. Therefore, the apparatus is redrawn as given below:
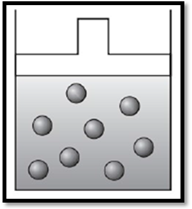
Explanation of Solution
Given data is that the temperature increases from
Charles’s law is the
From Charles’s law we know that
According to Charles’s law, if temperature increases at constant pressure, then volume also increases. Therefore, the apparatus is redrawn as given below:
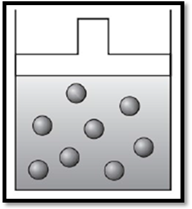
(b)
Interpretation:
The apparatus has to be redrawn if the pressure is increased from
Concept introduction:
Boyle’s law:
At fixed temperature, the volume of a fixed amount of gas is inversely proportional to the pressure exerted by the gas.
(b)
Answer to Problem 8.26UKC
According to Boyle’s law, if pressure increases at constant temperature, then volume decreases. Therefore, the apparatus is redrawn as given below:
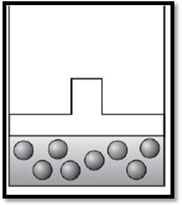
Explanation of Solution
Given data is that the pressure is increased from
Boyle’s law is the law which relates pressure and volume at a constant temperature and number of molecules.
From Boyle’s law:
According to Boyle’s law, if pressure increases at constant temperature, then volume decreases. Therefore, the apparatus is redrawn as given below:
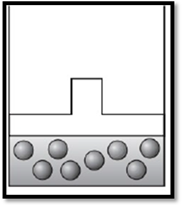
(c)
Interpretation:
The apparatus has to be redrawn if the temperature is decreased from
Concept introduction:
General Gas Law:
Combining Charles’s law and Boyle’s law we get the General gas law or combined gas law.
(c)
Answer to Problem 8.26UKC
The volume remains unchanged and the apparatus is redrawn as given below:
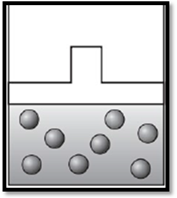
Explanation of Solution
Given data is that the temperature is decreased from
Combined gas law is proposed by combining the Boyle’s law and Charle’s law and is given by:
Therefore, the volume remains unchanged and the apparatus is redrawn as given below:
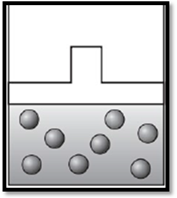
Want to see more full solutions like this?
Chapter 8 Solutions
Study Guide And Full Solutions Manual For Fundamentals Of General, Organic, And Biological Chemistry
- A sample of 8.00 mol of gas in a 10.00 L container is at 45.0 °C. What is the pressure (in atm) of the gas?arrow_forwardHow is Kw defined, and what is its numerical value at 25 °C (298 K)?arrow_forwardIf 18.0 g of O2 gas has a temperature of 350 K and a pressure of 550 mmHg, what is its volume?arrow_forward
- The vapour pressure of water at blood temperature is 47 Torr. What is the partial pressure of dry air in our lungs when the total pressure is 760 Torr?arrow_forwardA helium gas cylinder of the sort used to fill balloons has a volume of 0.180 m3 and a pressure of 150 X 105 Pa (150 atm) at 298 K (25 °C). How many moles of helium are in the tank? How many grams?arrow_forwardHeliox is a helium‑oxygen mixture that may be used in scuba tanks for divers working at great depths. It is also used medically as a breathing treatment. A 5.25 L tank holds helium gas at a pressure of 1734 psi. A second 5.25 L tank holds oxygen at a pressure of 461.0 psi. The two gases are mixed in a 5.25 L tank. If the temperature remains the same throughout the process, what is the pressure of the gas mixture in the tank? Assume ideal gas behavior.arrow_forward
- Nitric acid can be produced by the reaction ofgaseous nitrogen dioxide with water.3 NO2(g) + H2O(ℓ) −→2 HNO3(ℓ) + NO(g)If 956 L of NO2 gas react with water, whatvolume of NO gas will be produced? Assume the gases are measured under the sameconditionsarrow_forwardAccording to Dalton’s law, what is the partial pressure of amixture of gases? What is water vapor pressure?arrow_forwardDetermine and report the pressure of collected hydrogen gas in atmospheres. Report this result to one more digit than allowed by applying the rules of significant figures. (1 atm = 760 mm Hg, an exact conversion factor.) Determine the moles of hydrogen collected by substituting the appropriate quantities into the ideal gas equation and solving. Report this result to one more digit than allowed by applying the rules of significant figures. Notes: the gas temperature is the same as the listed temperature; R = 0.082057 L-atm/mol-K. Be mindful of units. Determine the experimental atomic mass of Cu. Round this result to the correct number of significant figures. Determine the percent relative error (= ????????????−?h????????? ? 100). The molar mass of Cu is 63.546 g/mol. Mass of copper before electrolysis 6.008 g Mass of copper after electrolysis 5.860 g Volume of H2 collected 58.9 mL Pressure of H2, PH2 720.8 mm Hg Temperature 24.0 oCarrow_forward
- A typical room is 4.0 m long, 5.0 m wide, and 2.5 m high. What is the total mass of the oxygen in the room assuming that the gas in the room is at STP and that air contains 21% oxygen and 79% nitrogen?arrow_forward15.0 L of an ideal gas at 298 K and 3.36 atm are heated to 383 K with a new pressure of 6.05 atm. What is the new volume in liters?arrow_forwardA bicycle pump contains 0.682 liters of air at 99.3 kpa . If the handle is pressed down decreasing the volume of the inside air to 0.151 L, what is the pressure inside the pump ? Assume temperature is constantarrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning



