
FUND.OF GEN,ORG.+...-MOD.ACCESS>CUSTOM<
17th Edition
ISBN: 9781323470473
Author: McMurry
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 8.29UKC
Three bulbs, two of which contain different gases and one of which is empty, are connected as shown in the following drawing:
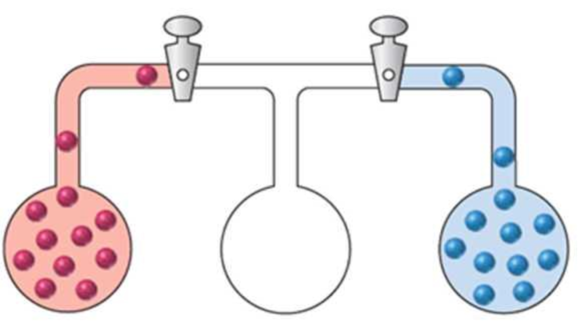
Redraw the apparatus to represent the gases after the stopcocks are opened and the system is allowed to come to equilibrium.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
During transfer of liquids using a micropipette:
What would it mean if the mean is very close to the expected value but your standard deviation is high ?
A sample of nitrogen gas occupies a volume of 2.00 L at 756 mm Hg and 0.00° C. Thevolume increases by 2.00 L and the temperature decreases to 137 K. What is the finalpressure exerted on the gas?
A cylinder of gas at room temperature (20 degrees Celsius) has a pressure P1. To what temperature in degrees Celsius would the temperature have to be increased for the pressure to be 1.2p1?
Chapter 8 Solutions
FUND.OF GEN,ORG.+...-MOD.ACCESS>CUSTOM<
Ch. 8.2 - Would you expect the boiling points to increase or...Ch. 8.2 - Prob. 8.3PCh. 8.2 - Identify the intermolecular forces (dipoledipole,...Ch. 8.4 - Prob. 8.5PCh. 8.4 - Prob. 8.6PCh. 8.4 - What evidence is there that global warming is...Ch. 8.4 - Prob. 8.2CIAPCh. 8.4 - Prob. 8.7PCh. 8.4 - Prob. 8.8KCPCh. 8.5 - Prob. 8.3CIAP
Ch. 8.5 - Prob. 8.4CIAPCh. 8.5 - Prob. 8.5CIAPCh. 8.5 - Prob. 8.9PCh. 8.5 - Prob. 8.10PCh. 8.6 - Prob. 8.11PCh. 8.7 - Prob. 8.12PCh. 8.8 - Prob. 8.13PCh. 8.8 - Prob. 8.14KCPCh. 8.9 - Prob. 8.15PCh. 8.10 - Prob. 8.16PCh. 8.10 - Prob. 8.17PCh. 8.10 - Prob. 8.18KCPCh. 8.11 - Prob. 8.19PCh. 8.11 - Prob. 8.20PCh. 8.11 - Prob. 8.21PCh. 8.11 - Prob. 8.22KCPCh. 8.14 - How much heat in kilocalories is required to (a)...Ch. 8.14 - Prob. 8.24PCh. 8.14 - Compare the Hvap values for water, isopropyl...Ch. 8.14 - What is a supercritical fluid?Ch. 8.14 - What are the environmental advantages of using...Ch. 8.14 - Prob. 8.8CIAPCh. 8 - Prob. 8.26UKCCh. 8 - Assume that you have a sample of gas at 350 K in a...Ch. 8 - Prob. 8.28UKCCh. 8 - Three bulbs, two of which contain different gases...Ch. 8 - Prob. 8.30UKCCh. 8 - The following graph represents the heating curve...Ch. 8 - Prob. 8.32UKCCh. 8 - Prob. 8.33UKCCh. 8 - Prob. 8.34APCh. 8 - Identify the predominant intermolecular force in...Ch. 8 - Dimethyl ether (CH3OCH3) and ethanol (C2H5OH) have...Ch. 8 - Prob. 8.37APCh. 8 - Prob. 8.38APCh. 8 - Prob. 8.39APCh. 8 - What are the four assumptions of the...Ch. 8 - Prob. 8.41APCh. 8 - Prob. 8.42APCh. 8 - Prob. 8.43APCh. 8 - Prob. 8.44APCh. 8 - Prob. 8.45APCh. 8 - Prob. 8.46APCh. 8 - Which assumptions of the kinetic-molecular theory...Ch. 8 - Prob. 8.48APCh. 8 - Prob. 8.49APCh. 8 - The use of CFCs as refrigerants and propellants in...Ch. 8 - Prob. 8.51APCh. 8 - Prob. 8.52APCh. 8 - Prob. 8.53APCh. 8 - Prob. 8.54APCh. 8 - Prob. 8.55APCh. 8 - Prob. 8.56APCh. 8 - Prob. 8.57APCh. 8 - Prob. 8.58APCh. 8 - Which assumptions of the kinetic-molecular theory...Ch. 8 - Prob. 8.60APCh. 8 - Prob. 8.61APCh. 8 - Prob. 8.62APCh. 8 - Prob. 8.63APCh. 8 - Prob. 8.64APCh. 8 - Prob. 8.65APCh. 8 - Prob. 8.66APCh. 8 - Prob. 8.67APCh. 8 - Prob. 8.68APCh. 8 - Prob. 8.69APCh. 8 - Prob. 8.70APCh. 8 - Prob. 8.71APCh. 8 - What is the mass of CH4 in a sample that occupies...Ch. 8 - Prob. 8.73APCh. 8 - Prob. 8.74APCh. 8 - Prob. 8.75APCh. 8 - Prob. 8.76APCh. 8 - Prob. 8.77APCh. 8 - Prob. 8.78APCh. 8 - Prob. 8.79APCh. 8 - Prob. 8.80APCh. 8 - Prob. 8.81APCh. 8 - Prob. 8.82APCh. 8 - Prob. 8.83APCh. 8 - Prob. 8.84APCh. 8 - Prob. 8.85APCh. 8 - Prob. 8.86APCh. 8 - Prob. 8.87APCh. 8 - Prob. 8.88APCh. 8 - Prob. 8.89APCh. 8 - Prob. 8.90APCh. 8 - Prob. 8.91APCh. 8 - Prob. 8.92APCh. 8 - Prob. 8.93APCh. 8 - Prob. 8.94APCh. 8 - Patients with a high body temperature are often...Ch. 8 - Prob. 8.96APCh. 8 - List three kinds of crystalline solids, and give...Ch. 8 - The heat of fusion of acetic acid, the principal...Ch. 8 - Prob. 8.99APCh. 8 - Prob. 8.100CPCh. 8 - Prob. 8.101CPCh. 8 - Prob. 8.102CPCh. 8 - Prob. 8.103CPCh. 8 - Prob. 8.104CPCh. 8 - Prob. 8.105CPCh. 8 - Prob. 8.106CPCh. 8 - Prob. 8.107CPCh. 8 - Prob. 8.108CPCh. 8 - Ethylene glycol, C2H6O2, has one OH bonded to each...Ch. 8 - Prob. 8.110CPCh. 8 - Prob. 8.111GPCh. 8 - Prob. 8.112GPCh. 8 - Prob. 8.113GPCh. 8 - Prob. 8.114GPCh. 8 - Prob. 8.115GP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Why are BSL-4 suits pressurized? Why not just wear tough regular suits?
Microbiology with Diseases by Body System (5th Edition)
How is a mailing address analogous to biologys hierarchical classification system?
Campbell Biology in Focus
Where is transitional epithelium found and what is its importance at those sites?
Anatomy & Physiology
An example of a frictional force doing positive work.
Physics (5th Edition)
In cats, tortoiseshell coat color appears in females. A tortoiseshell coat has patches of dark brown fur and pa...
Genetic Analysis: An Integrated Approach (2nd Edition)
88. Suppose that one of the people in Exercise 87 has twice the mass of the other. How far does each person sli...
Conceptual Physical Science (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Gas moves from an area of ________ partial pressure to an area of ________ partial pressure. low; high low; low high; high high; lowarrow_forwardInterpret the following ABG results: PH 7.48, PAC02 50 mm Hg, HCO3- 32 mEq/L: Interpret the following ABG results: PH 7.23, PAC02 40 mm Hg, HCO3- 19 mEq/L: Interpret the following ABG results: PH 7.48, PAC02 18 mm Hg, HCO3- 13 mEq/L:arrow_forward
Recommended textbooks for you
 Cardiopulmonary Anatomy & PhysiologyBiologyISBN:9781337794909Author:Des Jardins, Terry.Publisher:Cengage Learning,
Cardiopulmonary Anatomy & PhysiologyBiologyISBN:9781337794909Author:Des Jardins, Terry.Publisher:Cengage Learning, Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage LearningLifetime Physical Fitness & WellnessHealth & NutritionISBN:9781337677509Author:HOEGERPublisher:Cengage
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage LearningLifetime Physical Fitness & WellnessHealth & NutritionISBN:9781337677509Author:HOEGERPublisher:Cengage



Cardiopulmonary Anatomy & Physiology
Biology
ISBN:9781337794909
Author:Des Jardins, Terry.
Publisher:Cengage Learning,

Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning


Lifetime Physical Fitness & Wellness
Health & Nutrition
ISBN:9781337677509
Author:HOEGER
Publisher:Cengage
Microbial Nutrition and Growth; Author: Scientist Cindy;https://www.youtube.com/watch?v=rK3UkyWjkl8;License: Standard YouTube License, CC-BY