
Concept explainers
(a)
Interpretation:
The substitutive name for the given compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
The hydrocarbons that are attached to the longest chain are called substituents and they are written as prefix in alphabetical order.
Answer to Problem 8.5P
The substitutive name for the given compound is
Explanation of Solution
The given compound is
The substitutive name for the given compound is
(b)
Interpretation:
The substitutive name for the given compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
The hydrocarbons that are attached to the longest chain are called substituents and they are written as prefix in alphabetical order.
Answer to Problem 8.5P
The substitutive name for the given compound is
Explanation of Solution
The given compound is
The substitutive name for the given compound is
(c)
Interpretation:
The substitutive name for the given compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
The hydrocarbons that are attached to the longest chain are called substituents and they are written as prefix in alphabetical order.
Answer to Problem 8.5P
The substitutive name for the given compound is
Explanation of Solution
The given compound is shown below.
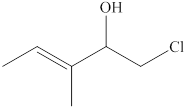
Figure 1
The given compound has a longest chain of five carbon atoms with a double bond on third carbon atom. It is an
The use of prefix
The substitutive name for the given compound is
(d)
Interpretation:
The substitutive name for the given compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
The hydrocarbons that are attached to the longest chain are called substituents and they are written as prefix in alphabetical order.
Answer to Problem 8.5P
The substitutive name for the given compound is
Explanation of Solution
The given compound is shown below.
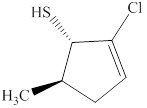
Figure 2
The given compound has a cyclic ring of five carbon atoms with a double bond on second carbon atom. It is an alkene, the suffix used for an alkene is –ene. Therefore, the parent name is
The naming of chiral center and geometric isomers are based on Cahn-Ingold-Prelog priority rules. If the priority assigned to each group attached to the chirality center in a molecule is in a clockwise direction, then it is the R-stereoisomer, and if this is counter-clockwise, then it is the S-stereoisomer. R and S-stereoisomer are mirror images of each other. Therefore, the name of this compound is
The substitutive name for the given compound is
(e)
Interpretation:
The substitutive name for the given compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
The hydrocarbons that are attached to the longest chain are called substituents and they are written as prefix in alphabetical order.
Answer to Problem 8.5P
The substitutive name for the given compound is
Explanation of Solution
The given compound is shown below.
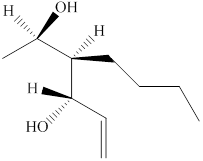
Figure 3
The given compound has a chain of six carbon atoms with a double bond on fifth carbon atom. It is an alkene, the suffix used for an alkene is –ene. Therefore, the parent name is
The naming of chiral center and geometric isomers are based on Cahn-Ingold-Prelog priority rules. If the priority assigned to each group attached to the chirality center in a molecule is in a clockwise direction, then it is the R-stereoisomer, and if this is counter-clockwise, then it is the S-stereoisomer. R and S-stereoisomer are mirror images of each other. Therefore, the name of this compound is
The substitutive name for the given compound is
(f)
Interpretation:
The substitutive name for the given compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
The hydrocarbons that are attached to the longest chain are called substituents and they are written as prefix in alphabetical order.
Answer to Problem 8.5P
The substitutive name for the given compound is
Explanation of Solution
The given compound is shown below.
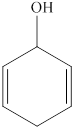
Figure 4
It consists of a cyclic ring of six carbon atoms. Therefore, the parent name is cyclohexa. There are two double bonds present at the second and fifth carbon atom of the ring. Also, one hydroxyl group is attached at the first carbon atom. Therefore, the name of this compound is
The substitutive name for the given compound is
(g)
Interpretation:
The substitutive name for the given compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
The hydrocarbons that are attached to the longest chain are called substituents and they are written as prefix in alphabetical order.
Answer to Problem 8.5P
The substitutive name for the given compound is
Explanation of Solution
The given compound is shown below.
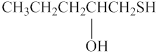
Figure 5
It consists of a chain of five carbon atoms with hydroxyl group attached at the second carbon atom. The alcohols contain hydroxyl
The substitutive name for the given compound is
(h)
Interpretation:
The substitutive name for the given compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
The hydrocarbons that are attached to the longest chain are called substituents and they are written as prefix in alphabetical order.
Answer to Problem 8.5P
The substitutive name for the given compound is
Explanation of Solution
The given compound is shown below.
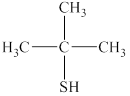
Figure 6
It consists of a chain of three carbon atoms with thiol group attached at the second carbon atom. One methyl group is attached at the second carbon atom. Therefore, the name of this compound is
The substitutive name for the given compound is
Want to see more full solutions like this?
Chapter 8 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- The functional group of butanone is:(a) Carboxyl(b) ketonic(c) aldehydic(d) alcoholicarrow_forwardDraw the condensed formulas for each of the following compounds:(a) dipropyl ether(b) 2,2-dimethyl-3-hexanol(c) 2-ethoxybutanearrow_forwardAlcohols can be produced by the hydration of:(a) Alkenes(b) alkynes(c) alkanes(d) acidsarrow_forward
- (B) Draw a structural diagram for each of the following organic compounds: (a) 2-ethyl-4-methyl-2-pentanol (b) 1,2-ethandiol (c) 1,3-dimethylbenzenearrow_forwardIdentify which of the following statement(s) is/are true. (a) A functional group is a group of atoms in an organic molecule that undergoes a predictable set of chemical reactions. (b) The functional group of an alcohol, an aldehyde, and a ketone have in common the fact that each contains a single oxygen atom. (c) A primary alcohol has one -OH group, a secondary alcohol has two -OH groups, and a tertiary alcohol has three -OH groups. (d) There are two alcohols with the molecular formula C3H8O. (e) There are three amines with the molecular formula C3H9N.arrow_forwardA certain hydrocarbon has a molecular formula of C5H8. Which of the following is not a structural possibility for this hydrocarbon: (d) It contains an alkyne O It contains one ring and one double bond (c) It contains two double bonds and no rings O (b) It contains one ring and no double bondsarrow_forward
- (a) The compound given below had the following IUPAC name and structural formula dibromocyclopentane C3H6CHBrCHBr (i) What type of isomerism is possible in the organic compound? (ii) Draw all the pairs of possible isomers and name them.arrow_forward(b) Differentiate between bioethanol and biodiesel.arrow_forwardWhat are the correct names for each compound.arrow_forward
- TRUE OR FALSE (a) A functional group is a group of atoms in an organic molecule that undergoes a predictable set of chemical reactions. (b) The functional group of an alcohol, an aldehyde, and a ketone have in common the fact that each contains a single oxygen atom. (c) A primary alcohol has one —OH group, a secondary alcohol has two —OH groups, and a tertiary alcohol has three —OH groups. (d) There are two alcohols with the molecular formula C3H8O. (e) There are three amines with the molecular formula C3H9N. (f) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (g) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (h) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic…arrow_forward(b) NABH, CH3 COCH,CH3 CH3CH2OH (c)arrow_forwardWrite an equation for each of the following reactions. Use molecular and structural formulas and classify the reaction as combustion, addition, substitution, hydrogenation, or hydration. (a) Octane burns with oxygen gas (b) 2-methyl-1-pentene reacts with hydrogen.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning


