
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8.7, Problem 16CC
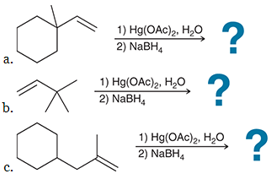
Predict the product for each reaction, and predict the products if an acid-catalyzed hydration had been performed rather than an oxymercuration - demercuration:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Choose the set of letters corresponding to the BEST answer.
-has four equivalent acidic hydrogens
-gives an iodoform upon reaction with I2/KI in NaOH
-a non-enolizable carbonyl compound
-yields 2-methylbutane when reacted with Zn(Hg) in HCl
name the major product for the reaction of 4-methyl-1-pentyne with the following reagents
⦁ H2/Li or Na/NH3 (liq) at -78 degrees C
dedcue the major organic product from the following reations. be ablw to draw hydration halogenation hydrohalogenation, halohydrin formation, oxymercuratiom demercuration qnd hydroboration oxidation.
Chapter 8 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Ch. 8.3 - Provide a systematic name for each of the...Ch. 8.3 - Prob. 2CCCh. 8.3 - Prob. 3CCCh. 8.3 - Prob. 4CCCh. 8.5 - Prob. 5CCCh. 8.5 - Prob. 6CCCh. 8.5 - Prob. 1LTSCh. 8.5 - Prob. 7PTSCh. 8.5 - Prob. 8ATSCh. 8.5 - Prob. 9CC
Ch. 8.5 - Prob. 2LTSCh. 8.5 - Prob. 10PTSCh. 8.5 - Prob. 11ATSCh. 8.6 - Prob. 12CCCh. 8.6 - Prob. 13CCCh. 8.6 - Prob. 3LTSCh. 8.6 - Prob. 14PTSCh. 8.6 - Prob. 15ATSCh. 8.7 - Predict the product for each reaction, and predict...Ch. 8.7 - Prob. 17CCCh. 8.8 - Prob. 18CCCh. 8.8 - Prob. 19CCCh. 8.8 - Prob. 4LTSCh. 8.8 - Prob. 20PTSCh. 8.8 - Prob. 21ATSCh. 8.9 - Prob. 5LTSCh. 8.9 - Prob. 22PTSCh. 8.9 - Prob. 23ATSCh. 8.10 - Prob. 24CCCh. 8.10 - Prob. 6LTSCh. 8.10 - Prob. 25PTSCh. 8.10 - Prob. 26ATSCh. 8.10 - Prob. 27ATSCh. 8.11 - Prob. 7LTSCh. 8 - Prob. 47PP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict the products of the following reaction. Include stereochemistry when necessary. For reactions with more than one step show the product formed after each step.arrow_forwardPredict the major products of the following reactions. o@xylene + H2 (1000 psi, 100 °C, Rh catalyst)arrow_forwardWhat products are obtained by hydration of the symmetrical alkyne shown below? Show the mechanism.arrow_forward
- Show how Wittig reactions might be used to synthesize the following compounds. Ineach case, start with an alkyl halide and a ketone or an aldehyde.(a) Ph¬CH“C(CH3 )2arrow_forwardFor each compound, give the product(s) expected from (1) HgSO4>H2SO4@catalyzedhydration and (2) hydroboration–oxidation.(a) hex-1-ynearrow_forwardPredict the products of the following reactions. Include stereochemistry when necessary. For reactions with more than one step show the product formed after each step.arrow_forward
- For each compound, give the product(s) expected from (1) HgSO4>H2SO4@catalyzedhydration and (2) hydroboration–oxidation.(a) hex-1-yne (b) hex-2-ynearrow_forwardIdentify all products that are expected for the following reaction.arrow_forwardPredict the major products of the following reactions, including stereochemistry whereappropriate.(a) 2,2-dimethyloxirane + H+ >H218O (oxygen-labeled water)arrow_forward
- Predict the product of the given compound when it react/s to the following reagents. Show the arrow-pushing mechanism a. xs O2arrow_forwardThe base-promoted rearrangement of an a-haloketone to a carboxylic acid, known as the Favorskii rearrangement, is illustrated by the conversion of 2-chlorocyclohexanone to cyclopentanecarboxylic acid. Jo Han Sow La NaOH, THF O Nat H3O+ OH NaOH, THF The mechanism involves the following 5 steps: -ō cyclopropanone intermediate 1. Abstraction of a proton to form enolate anion 1; 2. Formation of a cyclopropanone intermediate 2 with expulsion of chloride ion; 3. Addition of hydroxide ion to form tetrahedral intermediate 3; 4. Collapse of the tetrahedral intermediate and breakage of the three-membered ring to form carbanion intermediate 4; 5. Proton transfer to form the rearranged carboxylic acid. For the following reaction, draw the reaction out on paper, and then draw the structure of cyclopropanone intermediate 2 in the window. 1. NaOH, THF 2. H3O* OHarrow_forwardPredict the expected product for each reaction and provide IUPAC name for the correct starting material to yield the desired epoxide:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY