
(a)
Interpretation: The hydroboration-oxidation reaction product of alkene is to be interpreted.
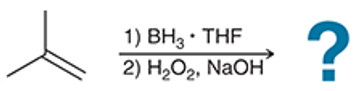
Concept introduction:
Addition reactions are the
(b)
Interpretation: The hydroboration-oxidation reaction product of alkene is to be interpreted.
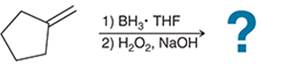
Concept introduction:
Addition reactions are the chemical reactions that show the addition of a certain group or molecule to the unsaturated carbon atoms. Alkenes are the unsaturated hydrocarbon that contains at least one double bond between the carbon atoms. Therefore, they tend to give addition reactions. Some examples of addition reactions are acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc.
(c)
Interpretation: The hydroboration-oxidation reaction product of alkene is to be interpreted.
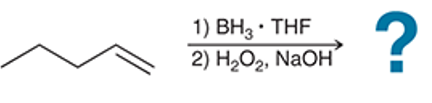
Concept introduction:
Addition reactions are the chemical reactions that show the addition of a certain group or molecule to the unsaturated carbon atoms. Alkenes are the unsaturated hydrocarbon that contains at least one double bond between the carbon atoms. Therefore, they tend to give addition reactions. Some examples of addition reactions are acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
- Give out chemical reactions of butadiene complexs Please answer breifly ASAParrow_forwardCalculate the standard free energy change (AG") for the transfer of electrons from succinate to ubiquinone: Succinate + ubiquinone O -2.7 kJ/mol relevant half-reactions and standard reduction potentials: fumarate + 2H* + 2 e™ H succinate ubiquinone + 2 H+ + 2 e² = O2.7 kJ/mol -8.7 kJ/mol O 14.7 kJ/mol succinate dehydrogenase O -6.0 kJ/mol Fumarate + ubiquinol ubiquinol E = 0.031 V ED - 0.045 V Atte 1 Harrow_forward1. Determine the product of (2red) resulting from the reaction of compound below with various reducing agents. 2. Determine the products of (2ox) resulting from the reaction of compound below with various oxidizing agents. 3. Determine the reduction products (3red) resulting from the reaction of 2ox compounds with various reducing agents. 4. Determine the oxidation products (3ox) resulting from the reaction of 2red compounds with various oxidizing agents.arrow_forward
- Compare the energetic favorability of MnO2 reduction by sulfide yielding S0 to the energetic favorability of MnO2 reduction by sulfide yielding SO42-.arrow_forwardWhat color will be produced if cyclohexene and toluene are reacted with sulfuric acid?arrow_forwardWhere does the strongest IMF occur in liquid ethanol?arrow_forward
- Why wouldn't this ruthenium catelyst, with two trans IMes ligands, be good at catalysing alkene metathesis?arrow_forward(a) What two components are needed to prepare para red by azo coupling? (b) What two components are needed to prepare alizarine yellow R?arrow_forwardWhat is the Noyori asymmetric reduction?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





