
Concept explainers
1. Use the following graph to diagram the energetics of a
To describe:
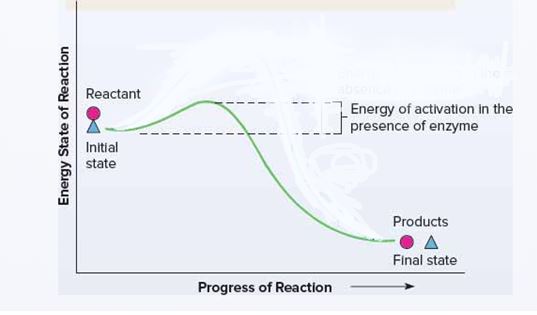
The energetics of a chemical reaction with and without an enzyme. To position reactants and products at appropriate points and to indicate the stages in the reaction and the energy levels.
Introduction:
Enzymes are remarkable example of protein biocatalyst that facilitates metabolic reactions.
Explanation of Solution
Enzymes increase the rate of a chemical reaction without becoming part of the products or being consumed in the reaction. Because of the free energy inherent in molecules, a reaction could occur spontaneously at some point even without an enzyme but at a very slow rate.
Energitics of a chemical reaction with an enzyme:
Energitics of a chemical reaction without an enzyme:
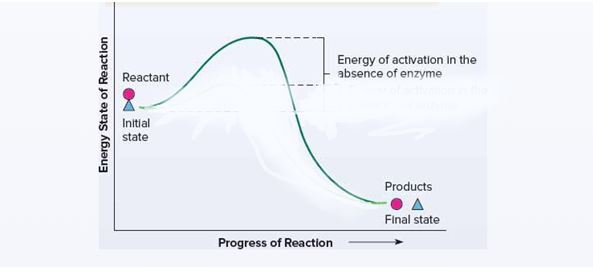
Enzymes increase the rate of a chemical reaction without becoming part of the products or being consumed in the reaction.
Want to see more full solutions like this?
Chapter 8 Solutions
Foundations in Microbiology
- A reducing chemical reaction. reduces the compound to a simpler form adds an electron to the substrate removes a hydrogen atom from the substrate is a catabolic reactionarrow_forwardIn the following graph: A represents the product. B represents the energy of activation when enzymes are present. C is the free energy difference between A and D. C is the energy of activation without enzymes. E is the difference in free energy between the reactant and the products.arrow_forwardWhich of the following comparisons or contrasts between endergonic and exergonic reactions is false? Endergonic reactions have a positive ?G and exergonic reactions have a negative ?G Endergonic reactions consume energy and exergonic reactions release energy Both endergonic and exergonic reactions require a small amount of energy to overcome an activation barrier Endergonic reactions take place slowly and exergonic reactions take place quickly.arrow_forward
- 1) Answer the following questions about a Vegetarian diet: a) List Pros and Cons of being on the diet. b) List any possible misconceptions that may have been verified or nullified by scientific articles for this diet. c) What biochemical pathway would this diet target?arrow_forward1. Looking at the graph you should be able to explain every activity of every enzyme. Please describe each one of them. 1A: Why is Temperature and pH so important? When is an enzyme able to work properlyarrow_forwardUse the following graph to diagram the energetics of a chemical reaction, with and without an enzyme. Be sure to position reactants and products at appropriate points and to indicate the stages in the reaction and the energy levels.arrow_forward
- 1. What are the differences classification of enzymes? List 5 classification of enzymes and cite their functions.arrow_forward1. DEMONSTRATE BY ILLUSTRATION HOW COUPLED REACTION IS UTILIZED IN THE TRANSFER OF ENERGY FROM THE SUN TO THE RAISING OF YOUR THUMB. 2. DID YOU SEE THE SIGNIFICANCE/CONNECTION OF THE LESSON IN YOUR LIFE? WRITE A BRIEF DESCRIPTIONarrow_forward1. What is the significance of the effect of enzyme concentration to enzyme activity in relation to the medical aspect? a. A person with fever cannot eat well and may suffer from indigestion b. A person with hyperacidicity experience vomiting upon taking milk c. Abnormal level of diagnostic enzymes indicate damage in the body tissues d. Too much hydrogen peroxide in the body forms free radicals if not decomposed by peroxidase 2. What is the relation of the effect of temperature on enzyme activity to the medical field? a. To explain the cause why a feverish person cannot eat b. To explain why drinking carbonated leaves a fizzing sensation in our tongue c. To explain the cause why milk should not be taken by hyperacidic individual d. To explain why bubbles are formed when cleaning wounds with hydrogen peroxide 3. What reaction was involved in the determination of Vitamin C content of fruits? a. Oxidation…arrow_forward
- One major difference between inorganic catalysts and organic catalysts is: A. Inorganic catalysts may not be compatible with biological parameters B. Organic catalysts are consumed during the chemical reaction C. Organic catalysts influence the activation energy of a reaction D. Inorganic catalysts decrease the probability a reaction will occurarrow_forward1. Why do we need to repeat the measurements of reaction of times ten times?arrow_forwardDescribe 3 different types of energy.arrow_forward
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning



