
a)
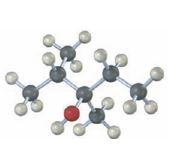
Interpretation:
The structures of
Concept introduction:
The hydroboration reaction takes place with syn stereochemistry and results in a non-Markovnikov addition of water to the double bond in the alkene. The resulting product has the hydroxyl group on the carbon with less number of alkyl substitutents.
The oxymercuration-demercuration reaction takes place with anti stereochemistry and results in Markovnokov addition of water to the double bond in the alkene. The resulting product has the hydroxyl group on the carbon with more number of alkyl substitutents.
To give:
The structures of alkenes that would yield the alcohol shown on hydration.
To state:
Among hydroboration-oxidation and oxymercuration-demercuration methods which method can be used to prepare the alcohol from alkenes.
b)
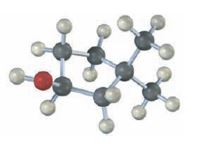
Interpretation:
The structures of alkenes that would yield the alcohol shown on hydration are to be given. Among hydroboration-oxidation and oxymercuration-demercuration, the method which can be used to prepare the alcohol also has to be stated.
Concept introduction:
The hydroboration reaction takes place with syn stereochemistry and results in a non-Markovnikov addition of water to the double bond in the alkene. The resulting product has the hydroxyl group on the carbon with less number of alkyl substitutents.
The oxymercuration-demercuration reaction takes place with anti stereochemistry and results in Markovnokov addition of water to the double bond in the alkene. The resulting product has the hydroxyl group on the carbon with more number of alkyl substitutents.
To give:
The structures of alkenes that would yield the alcohol shown on hydration.
To state:
Among hydroboration-oxidation and oxymercuration-demercuration, the method which can be used to prepare the alcohol.
Trending nowThis is a popular solution!

Chapter 8 Solutions
Organic Chemistry
- Show how to control the hydration of alkenes to give alcohols with either Markovnikov or anti-Markovnikov orientation, depending on the reagents.arrow_forwardThe hydration reaction of 1-hexene includes the addition of water across the double bond, adding an alcohol group to the more substituted carbon. Sulfuric acid is used as the catalyst for the reaction, which donates a hydrogen. The resulting product is a racemic mixture of 2-hexanol. A. Draw the alkene hydration reaction and its product(s) using information given above. B. Please include reaction mechanism with arrows showing all steps resulting in product formation.arrow_forwardAnswer the following questions about the mechanism for the acid-catalyzed hydration of an alkene: a. How many transition states are there? b. How many intermediates are there? c. Which step in the forward direction has the smallest rate constant?arrow_forward
- Draw the structure of two alkenes that would yield 1‑methylcyclohexanol when treated with Hg(OAc)2Hg(OAc)2 in water, then NaBH4NaBH4arrow_forwardAssign E or Z to the following alkenesarrow_forwardIdentify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.arrow_forward
- draw the two possible carbocations that can form when this alkene reacts with a strong acid (such as HBr or H3O+). of the two structures you drew, circle the more stable carbocationarrow_forwardProvide the major product for the reaction of the following starting material with H2O, H+. If more than one product is formed, list the major product first. If no reaction, draw the starting material.arrow_forwardDraw the major organic product formed by reaction of 2-hexyne with the following reagent: H2/ Lindlar catalyst. Consider E/Z stereochemistry of alkenes. In cases where there is more than one answer, just draw one. If no reaction occurs, draw the organic starting material.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

