
a)

Interpretation:
The product of the reaction shown if the reaction was conducted in DMSO with water is to be given. The complete mechanism of the reaction including appropriate regiochemistry and stereochemistry is also to be given.
Concept introduction:
The addition of halogens to
To give:
The product of the reaction shown if the reaction was conducted in DMSO with water and to give the complete mechanism of the reaction including appropriate regiochemistry and stereochemistry.
b)
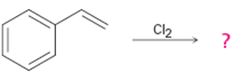
Interpretation:
The product of the reaction shown if the reaction was conducted in DMSO with water is to be given. The complete mechanism of the reaction including appropriate regiochemistry and stereochemistry is also to be given.
Concept introduction:
The addition of halogens to alkenes in the presence of aqueous DMSO results in the anti addition of the halohydrin, HOX, to the double bond. In the first step a cyclic halonium ion is formed by the attack of the double bond on the halogen. In the second step water attacks the halonium ion from the least shielded side to give an anti addition product. The addition obeys Markovnikov orientation. The negative part (OH) adds to the doubly bonded carbon atom which has more number of substituents.
To give:
The product of the reaction shown if the reaction was conducted in DMSO with water and to give the complete mechanism of the reaction including appropriate regiochemistry and stereochemistry.
c)
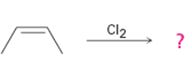
Interpretation:
The product of the reaction shown if the reaction was conducted in DMSO with water is to be given. The complete mechanism of the reaction including appropriate regiochemistry and stereochemistry is also to be given.
Concept introduction:
The addition of halogens to alkenes in the presence of aqueous DMSO results in the anti addition of the halohydrin, HOX, to the double bond. In the first step a cyclic halonium ion is formed by the attack of the double bond on halogen. In the second step water attacks the halonium ion from the least shielded side to give an anti addition product. The addition obeys Markovnokov orientation. The negative part (OH) adds to the doubly bonded carbon atom which has more number of substituents.
To give:
The product of the reaction shown if the reaction was conducted in DMSO with water and to give the complete mechanism of the reaction including appropriate regiochemistry and stereochemistry.
Trending nowThis is a popular solution!

Chapter 8 Solutions
Organic Chemistry
- Provide the mechanism and products for the acid-catalyzed epoxide opening reactions below, including appropriate stereochemistry.arrow_forwardUse your general knowledge of alkene chemistry to suggest a mechanism for the following reaction.arrow_forwardShow the stereochemistry of the epoxide (see Problem 11-31) you would obtain by formation of a bromohydrin from trans-2-butene, followed by treatment with base.arrow_forward
- In light of the fact that tertiary alkyl halides undergo spontaneous dissociation to yield a carbocation plus halide ion (see Problem 10-45), propose a mechanism for the following reaction.arrow_forwardThe HCo(CO)4complex can catalyze the isomerization of pure cis-2-butene to a mixture of cis-2-butene, trans-2-butene, and 1-butene. Propose a mechanism. Carefully show the process using wedges and dashes to indicate stereochemistry of each intermediate.arrow_forwardProvide the major product(s) for each reaction below and you may assume there is anexcess of each reagent for each reactionarrow_forward
