
(a)
Interpretation:
The areas for the pure solid, liquid and gas phases have to be labeled.
Concept Introduction:
Every pure substance that exists in all three phases has a characteristic phase diagram like below.
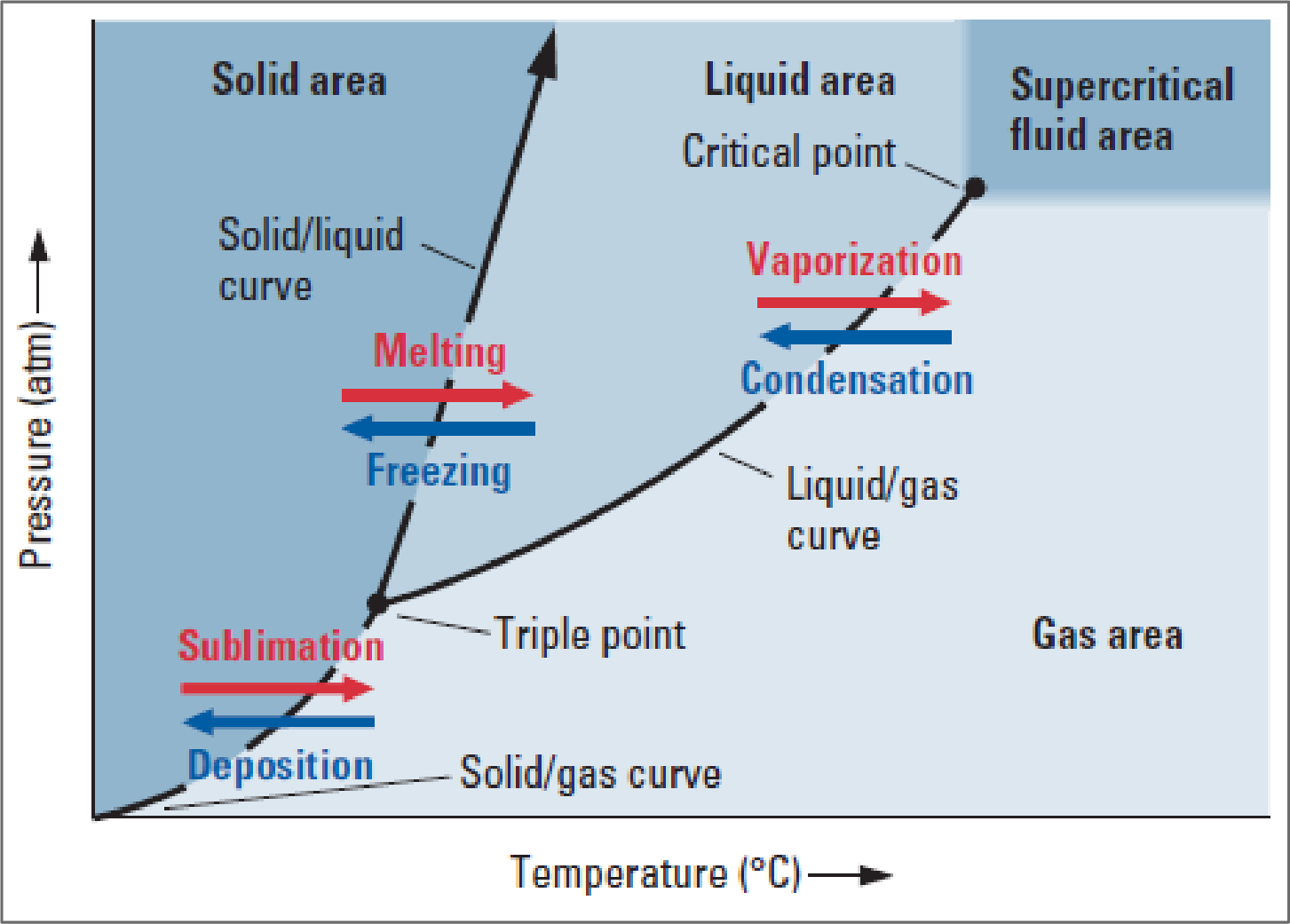
Figure 1
(b)
Interpretation:
A point on the diagram at which red phosphorus liquid and vapor are in equilibrium has to be labeled.
Concept Introduction:
Refer part (a).
(c)
Interpretation:
The reason why the solid red phosphorus cannot be melted in a container open to the atmosphere has to be explained.
(d)
Interpretation:
All the phase changes that occur sequentially when conditions change from point
Concept Introduction:
The temperature at which the vapour pressure of the liquid equals to the atmospheric pressure on the surface of the liquid is known as the boiling point.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Chemistry: The Molecular Science
- Define critical temperature and critical pressure. In terms of the kinetic molecular theory, why is it impossible for a substance to exist as a liquid above its critical temperature?arrow_forwardGiven this phase diagram and the data table, how do I arrive at the heat of vaporization (in kJ/mol)?arrow_forwardThe boiling point of ethyl ether was measured to be 10 °C at a base camp on the slopes of Mount Everest. as shown to determine the approximate atmospheric pressure at the camp.arrow_forward
- What is represented by A on the phase diagram?arrow_forwardIf the vacuum pump has sufficient capacity and is left on for an extended period of time, the liquid nitrogen will start to freeze. Explain why?arrow_forwardIn the phase diagram shown, segment AD corresponds to the conditions of temperature and pressure under which the solid and gas of the substance are in equilibrium. Select one:A)RealB) Falsearrow_forward
- Consider the phase diagram of carbon. If you heat a sample of graphite at a constant pressure of 1 atm, to what phase does it transform? If you compress a sample of graphite at 2000ºC, to what phase does it transform?arrow_forwardDetermine the normal boiling point of a liquid with vapor pressure 63.8 mmHg at 56.50°C, given that its molar heat of vaporization is 43.5 kJ/mol.arrow_forwardWhat solids are made up of large numbers of independent, constituent units, some of which attract while others repel.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





