
Interpretation:
The molecular geometry of
Concept introduction: In order to find the geometry of a molecule, follow the steps:
First, draw the Lewis dot structure of the molecule.
Find the number of electron pairs or domains around the central atom. Electron domain is the bond and lone pair of electrons around the central atom.
Use the VSEPR model to find the electron domain geometry.
According to VSEPR, the electron domains repel each other and arrange themselves as apart as possible in space.
Molecular geometry is the arrangement of bonded atoms. If no lone pairs are present, the molecular geometry is the same as the electron domain geometry.
The repulsion between two lone pairs is greater than that in case of lone pair–bond pair, which, in turn, is greater than the bond pair–bond pair repulsion.
Answer to Problem 1KSP
Correct answer: Option (c)
Explanation of Solution
Reason for the correct option:
Lewis dot structure of
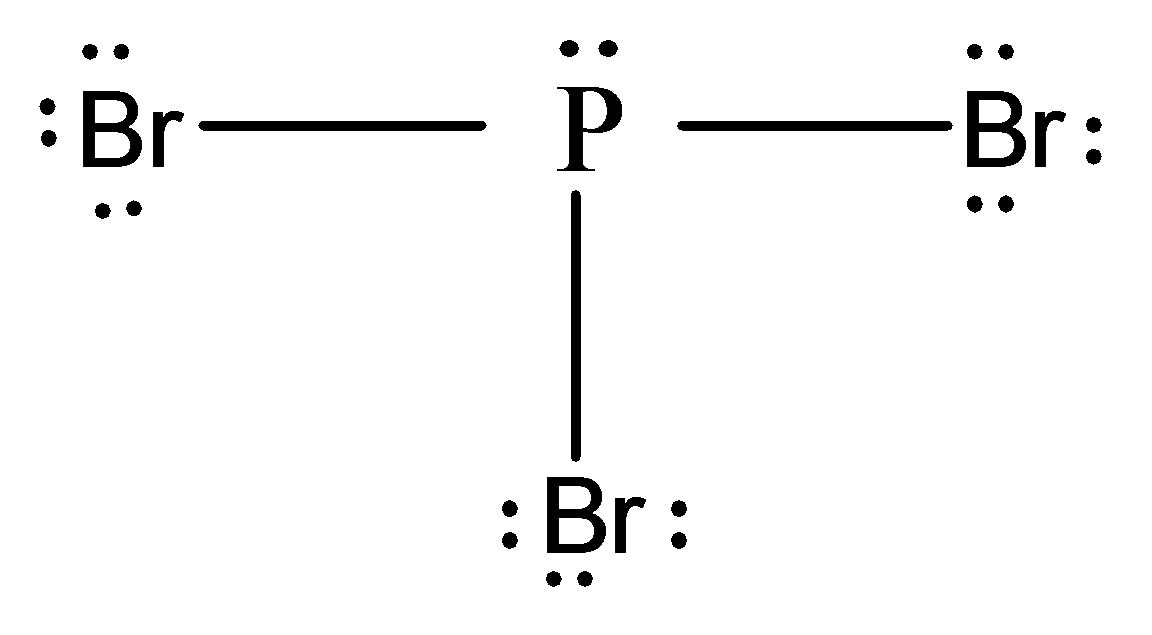
In this molecule, the phosphorus atom is bonded to three bromine atoms by single bonds and there is one lone pair on the phosphorus atom.
Thus, the electron domains around the phosphorus atom are four.
According to the VSEPR model, the four electron domains repel each other and arrange themselves in space in a tetrahedral shape.
However, in this case, one lone pair is present on the phosphorus atom.
Thus, the molecular geometry is trigonal pyramidal.
Hence, option (c) is correct.
Reasons for the incorrect options:
Option (a) is incorrect because a trigonal planar shape is obtained when there is no lone pair on the central atom.
Option (b) is incorrect because a tetrahedral shape is obtained when four bonds are present.
Option (d) is incorrect because a bent shape is obtained when there are two bonds present.
Option (e) is incorrect because a T-shape is obtained from the trigonal bipyramidal geometry with two lone pairs.
Hence, options (a), (b), (d), and (e) are incorrect.
Want to see more full solutions like this?
Chapter 9 Solutions
CHEMISTRY MCC CUSTOM W/CONNECT >CI<
- Sketch the resonance structures for the N2O molecule. Is the hybridization of the N atoms the same or different in each structure? Describe the orbitals involved in bond formation by the central N atom.arrow_forwardCompare the structure and bonding in CO2 and CO32 with regard to the OCO bond angle, the CO bond order, and the C atom hybridization.arrow_forwardGive the Lewis structure, molecular structure, and hybridization of the oxygen atom for OF2. Would you expect OF2 to be a strong oxidizing agent like O2F2 discussed in Exercise 61?arrow_forward
- 9.14 (a) Methane 1CH42 and the perchlorate ion 1ClO4- 2 are bothdescribed as tetrahedral. What does this indicate about theirbond angles? (b) The NH3 molecule is trigonal pyramidal,while BF3 is trigonal planar. Which of these molecules is flat?arrow_forwardPredict whether the N—O bonds in NO 3 are stronger or weaker than the N—O bonds in NO+.?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





