
Concept explainers
Explain in detail how you would distinguish between the following sets of compounds using the indicated method of spectroscopy.
(a)

(b)
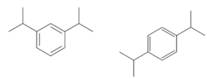
(c)
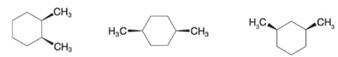
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
ORGANIC CHEMISTRY-WILEYPLUS ACCESS PKG.
Additional Science Textbook Solutions
Introductory Chemistry (5th Edition) (Standalone Book)
Chemistry: A Molecular Approach (4th Edition)
Chemistry: A Molecular Approach
Chemistry
Chemistry
Introductory Chemistry (6th Edition)
- Propose a structural formula for the analgesic phenacetin, molecular formula C10H13NO2, based on its 1H-NMR spectrum.arrow_forwardThe following 1H NMR peaks were recorded on a spectrometer operating at 200 MHz. Convert each into δ units. (a) CHCl3; 1454 Hz (b) CH3Cl; 610 Hz (c) CH3OH; 693 Hz (d) CH2Cl2; 1060 Hzarrow_forwardIdentify the compound with molecular formula C6H10O that gives the following DEPT 13C NMR spectrum:arrow_forward
- Which of the four structures is consistent with the 1H-NMR spectrum shown below? t, 2H 5 (a) (c) 4 ход ngx 3 PPM 2 tq, 2 H elle (b) (d) s. 9H t, 3 H 0arrow_forwardDescribe the 1H NMR spectrum of each compound. State how many NMR signals are present, the splitting pattern for each signal, and the approximate chemical shift.arrow_forwardSketch the 1H NMR spectra of the following compounds.arrow_forward
- Following is the 1H-NMR spectrum of compound O, molecular formula C7H12. Compound O reacts with bromine in carbon tetrachloride to give a compound with the molecular formula C7H12Br2. The 13C-NMR spectrum of compound O shows signals at d 150.12, 106.43, 35.44, 28.36, and 26.36. Deduce the structural formula of compound O.arrow_forwardThere are four esters with molecular formula C4H8O2. How can they be distinguished by 1H NMR?arrow_forwardThe 1H NMR spectrum of 1,2-dimethoxyethane (CH3OCH2CH2OCH3) recorded on a 300 MHz NMR spectrometer consists of signals at 1017 Hz and 1065 Hz downeld from TMS. (a) Calculate the chemical shift of each absorption. (b) At what frequency would each absorption occur if the spectrum were recorded on a 500 MHz NMR spectrometer?arrow_forward
- The 1H-NMR spectrum of compound B,C7H14O , consists of the following signals: δ 0.9 (t, 6H), 1.6 (sextet, 4H), and 2.4 (t, 4H). Draw the structural formula of compound B.arrow_forward(a) Draw all six isomers of formula C4H8 (including stereoisomers).(b) For each structure, show how many types of H would appear in the proton NMR spectrum.(c) For each structure, show how many types of C would appear in the 13C NMR spectrum.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

