
Concept explainers
Interpretation:
The series of equations for the preparation of each of the given compound from the designated starting material using suitable organic or inorganic reagents is to be written.
Concept introduction:
The conjugate base of alkyne acts as a good nucleophile. On reaction with primary
In hydration of alkynes, a molecule of water gets added to a molecule of alkyne.Hydrogen atom gets attached to the less substituted triple bonded carbon atom, and hydroxyl groupgetsbonded to the more substituted triple bonded carbon atom and forms a
Geminal and vicinal dihalides, on reaction with a strong base, undergo double dehydrohalogenation and form alkynes.
On halogenation, alkynes form vicinal dihalides and, on reaction with hydrogen halides, they form geminal dihalides in two steps.
Primary alcohol can be converted to primary alkyl bromide by reaction with sodium bromide in acidic condition.
Answer to Problem 36P
Solution:
a) 
b) 
c) 
d) 
e) 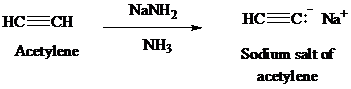

f) 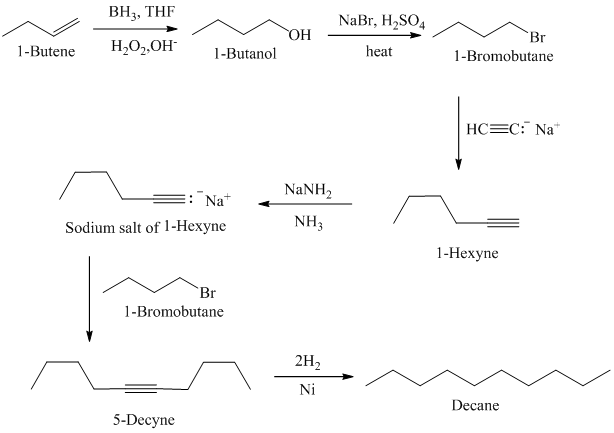
g) 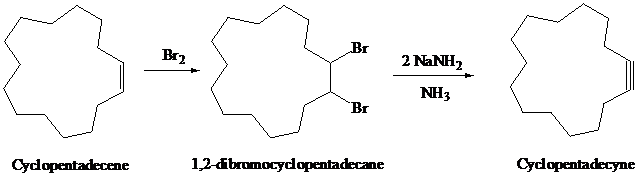
h) 
Explanation of Solution
a)
The
The synthesis of

b)
The
The synthesis of

c)
The
The synthesis of

d)
Acetylene on treatment with sodium amide, gives sodium salt of acetylene. The sodium salt of acetylene on treatment with ethyl bromide gives
The synthesis of

e)
Acetylene on treatment with sodium amide, gives sodium salt of acetylene. In presence of organoboron,
The synthesis of
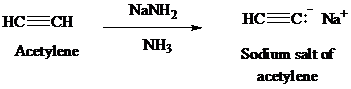

f)
In presence of organoboron,
The synthesis of

g)
Cyclopentadecene on halogenation with
The synthesis of
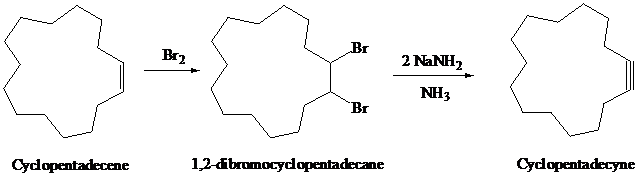
h) 
In presence of base sodium amide, the given alkyne gives sodium salt of alkyne. The sodium salt of alkyne on treatment with bromomethane adds one methyl group to alkyne, which on partial hydrogenation with Lindlar catalyst gives required cis alkene.
The synthetic route is as follows:

Want to see more full solutions like this?
Chapter 9 Solutions
ORGANIC CHEMISTRY 2 YEAR CONNECT ACCES
- Starting with benzene and using any other necessary reagents of your choice, what are the possible syntheses for the following compound?arrow_forwardShow by a series of equations how you could synthesize each of the following compounds from the indicated starting material and any necessary organic or inorganic reagents:(a) 2-Methylpropanoic acid from tert-butyl alcohol(b) 3-Methylbutanoic acid from tert-butyl alcohol(c) 3,3-Dimethylbutanoic acid from tert-butyl alcohol(d) HO2C(CH2)5CO2H from HO2C(CH2)3CO2H(g) 2,4-Dimethylbenzoic acid from m-xylene(h) 4-Chloro-3-nitrobenzoic acid from p-chlorotoluene(i) (Z)-CH3CH CHCO2H from propynearrow_forwardPredict the major organic products of the following reaction. You do not need to include stereochemistry in your answer. If no significant reaction will occur under the conditions shown, check the box under the drawing area instead. Note: for the purpose of this problem, simple acid-base reactions between the reagents should not be considered a significant reaction.arrow_forward
- Show how you can synthesize the following compounds starting with benzene, toluene, and alcohols containing no morethan four carbon atoms as your organic starting materials. Assume that para is the major product (and separable fromortho) in ortho, para mixtures.(a) pentan-1-amine (b) N-methylbutan-1-aminearrow_forwardName, draw and describe the organic product of the reaction between 2-methylbut-1-ene and H2O in the presence of H2SO4 and provide a clear rationale as to why this is the major product of the reaction and the minor product of the reaction.arrow_forwardGive me a clear handwritten answer with explanation...From cyclopropane, prepare 2,3-dimethylbutanearrow_forward
- Give the major organic product of each of the following reactions. Write NR if a reaction will not occur.arrow_forwardHow many possible monochlorination products are there for compound A? And what is its major monochlorination product (provide its structure)?arrow_forwardGive the organic product(s) formed in each of the following reactions. When necessary, draw a 3D representation for the product molecule showing its correct configuration.arrow_forward
- Like other electrophiles, carbocations add to alkenes to form new carbocations, which can then undergo substitution or elimination reactions depending on the reaction conditions. With this in mind, consider the following reactions of nerol, a natural product isolated from lemon grass and other plant sources. Treatment of nerol with TsOH forms α-terpineol as the major product, whereas treatment of nerol with chlorosulfonic acid, HSO3Cl, forms a constitutional isomer, α-cyclogeraniol. Write stepwise mechanisms for both processes. Each mechanism involves the addition of an electrophile—a carbocation—to a double bond.arrow_forwardLike other electrophiles, carbocations add to alkenes to form new carbocations, which can then undergo substitution or elimination reactions depending on the reaction conditions. With this in mind, consider the following reactions of nerol, a natural product isolated from lemon grass and other plant sources. Treatment of nerol with TsOH forms α-terpineol as the major product, whereas treatment of nerol with chlorosulfonic acid, HSO3Cl, forms a constitutional isomer, αcyclogeraniol. Write stepwise mechanisms for both processes. Each mechanism involves the addition of an electrophile— a carbocation—to a double bond.arrow_forwardPlease don't provide handwritten solution .... What is the major organic product from the following reaction?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
