
EBK GENERAL CHEMISTRY
11th Edition
ISBN: 9780133400588
Author: Bissonnette
Publisher: VST
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 9, Problem 45E
Match each of the lettered items on the left with an appropriate numbered item on the right. All the numbered items should be used, and some more than once. 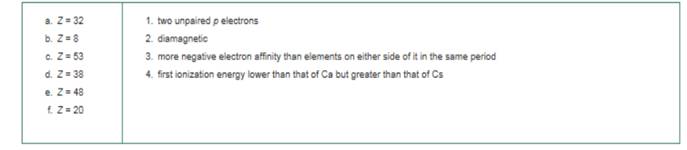
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The periodic table is the most important organizing principle in chemistry. There is a regular progression in the periodic table that reflects a similar regularity in the structures and properties of the different elements. If you know the properties of any one element in a group, you can make a good guess at the properties
of every other element in the same group and even the elements in neighboring groups. Familiarizing yourself with the structure and arrangement of the periodic table early on will help you later on in chemistry.
Part A
Classify the following elements as main group elements, transition metals, or inner transition metals.
Drag the appropriate elements to their respective bins.
► View Available Hint(s)
Main group elements
Submit
Ca Sn Fm Pd Zn S
Transition metals
Inner transition metals
Reset Help
What evidence is there that the colors observed in the flame tests are due to the metals, and not the nonmetals in the compounds tested?
Fill in the blanks using the other image.
Chapter 9 Solutions
EBK GENERAL CHEMISTRY
Ch. 9 - Prob. 1ECh. 9 - Suppose that lanthanum (Z = 57) were a newly...Ch. 9 - The following densities, in grams per cubic...Ch. 9 - The blowing melting points are in degrees Celsius....Ch. 9 - Mendeleev's periodic table did not preclude the...Ch. 9 - Prob. 6ECh. 9 - Prob. 7ECh. 9 - Concerning the incomplete seventh period of the...Ch. 9 - For each of the following pairs, indicate the atom...Ch. 9 - Indicate the smallest and the largest species...
Ch. 9 - Explain why the radii of atoms do not simply...Ch. 9 - The masses of individual atoms can be determined...Ch. 9 - Which is (a) the smallest atom in group 13; (b)...Ch. 9 - How would you expect the sizes of the hydrogen...Ch. 9 - Prob. 15ECh. 9 - Explain why the generalizations presented in...Ch. 9 - Among the following ions, several pairs are...Ch. 9 - Prob. 18ECh. 9 - All the isoelectronic species illustrated in the...Ch. 9 - Prob. 20ECh. 9 - Use principles established in this chapter to...Ch. 9 - Are there any atoms for which the second...Ch. 9 - Some electron affinities are negative quantities,...Ch. 9 - How much energy, in pules, must be absorbed to...Ch. 9 - How much energy, in kilojoules, is required to...Ch. 9 - Prob. 26ECh. 9 - The production of gaseous bromide ions from...Ch. 9 - Use ionization energies and electron affinities...Ch. 9 - The Naa ion and the Ne atom are isoelectronic. The...Ch. 9 - Prob. 30ECh. 9 - Compare the elements Al, Si, S, and Cl. a. Place...Ch. 9 - Compare the elements Na, Mg, O, and P. a. Place...Ch. 9 - Unpaired electrons are found in only one of the...Ch. 9 - Which of the following species has the greatest...Ch. 9 - Which of the following species would you expect to...Ch. 9 - Write electron configurations consistent with the...Ch. 9 - Must all atoms with an odd atomic number be...Ch. 9 - Neither Co24 nor Co24 has 4s electrons in its...Ch. 9 - Use ideas presented in this chapter to indicate...Ch. 9 - Arrange the following atoms in order of increasing...Ch. 9 - Arrange the following species in order of...Ch. 9 - For the following groups of elements, select the...Ch. 9 - Prob. 43ECh. 9 - Of the species Naa, Na, F, and F-, which has the...Ch. 9 - Match each of the lettered items on the left with...Ch. 9 - Prob. 46ECh. 9 - Which of the following ions are unlikely to be...Ch. 9 - Which of the following ions are likely to be found...Ch. 9 - Four atoms and/or ions are sketched below in...Ch. 9 - Prob. 50IAECh. 9 - In Mendeleev's time, indium oxide, which is 82.5%...Ch. 9 - Instead of accepting the atomic mass of indium...Ch. 9 - Refer to Figure 9-11 and explain why the...Ch. 9 - Explain why the third ionization energy of Li(g)...Ch. 9 - Prob. 55IAECh. 9 - Prob. 56IAECh. 9 - Studies done in 1880 showed that a chloride of...Ch. 9 - Assume that atoms are herd spheres, and use the...Ch. 9 - When sodium chloride is strongly heated in a...Ch. 9 - Use information from Chapters 8 and 9 to calculate...Ch. 9 - Refer only to the periodic table on the inside...Ch. 9 - Refer to the footnote on page 393. Then use values...Ch. 9 - Prob. 63IAECh. 9 - Prob. 64IAECh. 9 - The work functions for a number of metals are...Ch. 9 - The following are a few elements and their...Ch. 9 - Gaseous sodium atoms absorb quanta with the...Ch. 9 - A method for estimating electron affinities is to...Ch. 9 - We have seen that the wave functions of...Ch. 9 - In your own words, define the following terms (a)...Ch. 9 - Briefly describe each of the following ideas or...Ch. 9 - Explain the important distinctions between each...Ch. 9 - Prob. 73SAECh. 9 - Prob. 74SAECh. 9 - Prob. 75SAECh. 9 - Prob. 76SAECh. 9 - Prob. 77SAECh. 9 - An ion that is isoelectronic with Se2- is (a) S2-...Ch. 9 - Write electron configurations to show the first...Ch. 9 - Explain why the first ionization energy of Mg is...Ch. 9 - Prob. 81SAECh. 9 - Prob. 82SAECh. 9 - Find three pairs of elements that are out of order...Ch. 9 - Prob. 84SAECh. 9 - Prob. 85SAECh. 9 - Prob. 86SAECh. 9 - Prob. 87SAECh. 9 - Prob. 88SAECh. 9 - In multielectron atoms many of the periodic trends...Ch. 9 - Consider a nitrogen atom in the ground state and...Ch. 9 - Prob. 91SAECh. 9 - Describe how the ionization energies of the ions...Ch. 9 - Prob. 93SAECh. 9 - Prob. 94SAECh. 9 - When compared to a nonmetal of the same period, a...Ch. 9 - Prob. 96SAECh. 9 - Which of the following has a smaller radius than a...Ch. 9 - Prob. 98SAECh. 9 - The electrons lost when Fe ionizes to Fe2- are (a)...Ch. 9 - Prob. 100SAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- On a dry day, your hair flies apart when you brush it. How would you explain this?Match the words in the left column to the appropriate blanks in the sentences on the right.arrow_forwardThe vitamin niacin (nicotinic acid,C6H5NO2 ) can he isolated from a variety of natural sources, such as liver, yeast, milk, and whole grain. It also can be synthesized from commercially available materials. Which Source of nicotinic acid, from a nutritional view, is best for use in a multivitamin tablet? Why?arrow_forwardChemically active surfaces are seen on alumina-containing zeolites. Interpret this statement.arrow_forward
- W̲h̲y̲ ̲a̲r̲e̲ ̲s̲o̲m̲e̲ ̲m̲a̲t̲e̲r̲i̲a̲l̲s̲ ̲c̲o̲l̲o̲r̲e̲d̲ ̲a̲n̲d̲ ̲o̲t̲h̲e̲r̲s̲ ̲a̲r̲e̲ ̲n̲o̲t̲?̲ ̲H̲o̲w̲ ̲d̲o̲ ̲t̲h̲o̲s̲e̲ ̲m̲a̲t̲e̲r̲i̲a̲l̲s̲ ̲g̲e̲t̲ ̲t̲h̲e̲i̲r̲ ̲s̲p̲e̲c̲i̲f̲i̲c̲ ̲c̲o̲l̲o̲r̲?̲ ̲W̲h̲y̲ ̲a̲r̲e̲ ̲m̲e̲t̲a̲l̲s̲ ̲o̲p̲a̲q̲u̲e̲?̲arrow_forwardFor number 9 I need the words to match the meaning for all of themarrow_forward3. A prism is anle to differentiate the colors of the rainbow via refraction. The spectrometer uses a surface called a diffraction grating, which reflects light of various wavelengths at different angles. Give a real life example of an object or substance which acts as a diffracting grading, explain your reasoning.arrow_forward
- Analysis: What is an ion? Describe what happens to the electrons in an atom when they absorb energy (from the flame)? Why does this produce a bright-line emission spectrum (rather than a continuous spectrum)? In terms of atomic structure, why does each metal ion produce a different color flame? What difficulties are there in identifying metal ions with a flame test? How could you improve the specific identification of the metal ions during a flame test? If it can be determined from the flame test results, what are the identities of the two unknown liquids? What might be some practical (and exciting) applications of metal containing compounds that create different colors of light while burning? Explain.arrow_forwardThe following radial probability diagram could theoretically represent which orbital (s)? Select as many answers as are correct. Do not worry about the numerical values on the x/y axes. Just focus on the general shape of the function. r²R(r)² 0.6 0.5 0.4 0.3 0.2 0.1 0.0 0 4 8 12 16 20 24 28 32 36 r/ao 40arrow_forward1. The vertical columns in the periodic table are called _ halogens periods groups valence 2. Which of the following are not correctly paired? Na, alkali metal Br, halogen Ar, noble gas Sn, lanthanide 3. Which of the following elements belongs to the d-block? magnesium silver uranium galliumarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Periodic Properties of Elements | Chemistry | IIT-JEE | NEET | CBSE | Misostudy; Author: Misostudy;https://www.youtube.com/watch?v=L26rRWz4_AI;License: Standard YouTube License, CC-BY
Periodic Trends: Electronegativity, Ionization Energy, Atomic Radius - TUTOR HOTLINE; Author: Melissa Maribel;https://www.youtube.com/watch?v=0h8q1GIQ-H4;License: Standard YouTube License, CC-BY