
Draw all constitutional isomers having molecular formula C4H100. Classify each compound as a 1°, 2°, or 3° alcohol, or a symmetrical or unsymmetrical ether.
Interpretation:
To draw all the constitutional isomers for the molecular formula C4H10O and classify each compound as a 1°, 2°, or 3° alcohol, or a symmetrical or unsymmetrical ether.
Concept introduction:
Constitutional isomers also known as structural isomers. They are molecules that have the same chemical formula but their atoms are arranged differently. They are three main types of constitutional isomers.
a) Chain isomers – the carbon chain has a different order of carbon and hydrogen atoms.
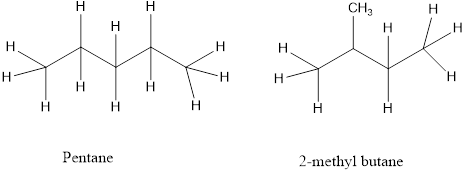
b) Position isomers- a functional group of the isomers is in a different location on the molecules.
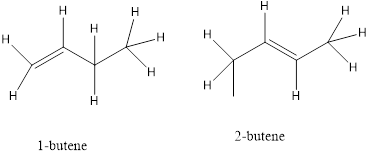
c) Functional isomers- the isomers contain a different functional group.
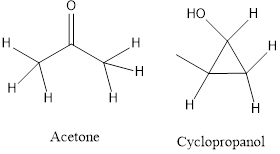
Answer to Problem 9.1P
Molecular formula C4H10O may be an alcohol and ether and seven isomers are shown below.
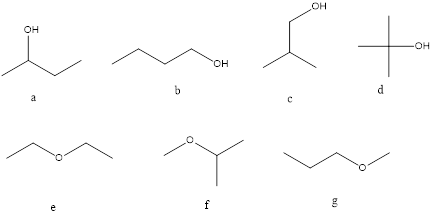
Strucutre,
A= 2o alcohol
B=1o alcohol
C=1o alcohol
D=3o alcohol
E=Symmetrical ether
F=Unsymmetrical ether
G=Unsymmetrical ether
Explanation of Solution
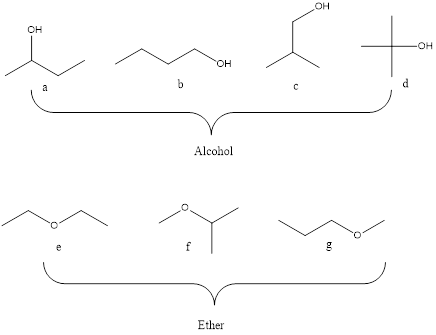
Alcohol are classified into 1o, 2o and 3o alcohol based on the number of the carbon atom bonded to carbon with OH functional group. Whereas the ether are symmetrical and unsymmetreical ethers. The ether that have two identical R groups is said to be symmetrical ether and having different R group is said to unsymmetrical ether.
Strucutre,
A= 2o alcohol
B=1o alcohol
C=1o alcohol
D=3o alcohol
E=Symmetrical ether
F=Unsymmetrical ether
G=Unsymmetrical ether
Thus seven constitutional isomers with functional groups alcohol and ether are obtained for the molecular formula C4H10O. And the structure,
A= 2o alcohol
B=1o alcohol
C=1o alcohol
D=3o alcohol
E=Symmetrical ether
F=Unsymmetrical ether
G=Unsymmetrical ether
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry (Looseleaf)
- why do some ethers with the same molecular formulas as their respective alcohols have a lower boiling point?arrow_forwardDraw and name using the IUPAC system all the constitutional isomers (thiols and sulfides) having molecular formula C4H10S. Classify each ether compound as symmetric or unsymmetric.arrow_forwardDraw structures for the four constitutional isomers of molecular formula C4H10O that contain an OH group. Give the IUPAC name for each alcohol.arrow_forward
- Identify each compound as an ether, hemiacetal, or acetal.arrow_forward1. What general conclusions can be drawn regarding the solubility of alcohols in water? Explain which of 1-pentanol and 1-heptanol will be more soluble in water?arrow_forwardAlthough diethyl ether and tetrahydrofuran are both four-carbon ethers, one compound is much more water soluble than the other. Predict which compound has higher water solubility and offer an explanation ?arrow_forward
- Draw and name all constitutionally isomeric alcohols with the molecular formula C4H10Oarrow_forwardWhat products are formed when each alcohol is oxidized with K 2Cr 2O 7? In some cases, no reaction occurs.arrow_forwardArrange these compounds in order of increasing boiling point (values in C are 42, 24, 78, and 118). (a) CH3CH2OH (b) CH3OCH3 (c) CH3CH2CH3 (d) CH3COOHarrow_forward
- Draw a condensed structure and a skeletal structure for each of the following:a. sec-butyl tert-butyl ether e. 5-(1-methylethyl)nonane i. 3-ethoxy-2-methylhexaneb. isoheptyl alcohol f. triethylamine j. 5-(1,2-dimethylpropyl)nonanec. sec-butylamine g. 4-(1,1-dimethylethyl)heptane k. 3,4-dimethyloctaned. isopentyl bromide h. 5,5-dibromo-2-methyloctane l. 5-isopentyldecanearrow_forwardPBr3 (phosphorus tribromide) converts alcohols into alkyl bromides. Define ?arrow_forwardTrue or False. Organic compounds synthesized in the laboratory have the same chemical and ohysical properties as those synthesized in living organisms. True or False. An alcohol, an aldehyde, and a ketone each contain a single oxygen atom. Multiole choice 1. Which of the following molecules contains a carbonyl group? A. 2-Propanol B. Diethyl ether C. 3,7-Dimethyloctanal D. 2-Methyl-1-butene 2. Methanol has all the following properties except? A. Colorless liquid B. Poisonous C. Boils below 100°C D. Does not hydrogen bondarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



