
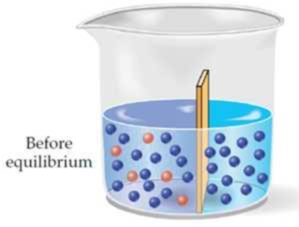
Assume that two liquids are separated by a semipermeable membrane, with pure solvent on the right side and a solution of a solute on the left side. Make a drawing that shows the situation after equilibrium is reached.
Interpretation:
The situation after equilibrium is reached when two liquids are separated by a semipermeable membrane has to be drawn.
Concept introduction:
Colligative properties such as vapor pressure, freezing point, and boiling point are affected by the presence of solute particles in a solution.
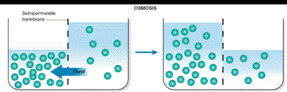
Osmosis is the passage of substances in true solution through a semipermeable membrane.
Particles such as starches and protein molecules are too large to pass the membrane.
The restriction of passage of large particles causes the smaller particles to pass more rapidly in the direction of higher concentrations, producing an osmotic pressure in confined liquids.
Explanation of Solution
Osmosis occurs when solution of different concentration are separated by a semipermeable membrane that allows solvent molecules to pass but blocks the passage of solute ions and molecules. Solvent flows from the more dilute side to the more concentrated side until sufficient osmotic pressure builds up and stops the flows. An effect similar to osmosis occurs when membranes of large pore size are used.
The situation after equilibrium is reached when two liquids are separated by a semipermeable membrane is drawn as
Want to see more full solutions like this?
Chapter 9 Solutions
Mastering Chemistry with Pearson eText -- Standalone Access Card -- for Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- What is the definition of solute concentration? A. The total amount of solute inside a cell B. The total amount of solution inside and outside a cell C. The total amount of solvent in a solution D. The total amount of solute dissolved in a solventarrow_forwardwhy does water move inward in a hypotonic solution?arrow_forwardfor 0.9% NaCl solution and 10% NaCl solution, which one has higher concentration of WATER?arrow_forward
- Is a solution highly concentrated if it is saturated? Is a solution saturated if it is highly concentrated?arrow_forwardWhich is NOT an appropriate factor in choosing solvents in a mixed solvent system? a. The two solvents must be inert. b. The two solvents must be immiscible. c. The solute must have varying solubility between the two solvents d. The solute must not react with the solventsarrow_forwardWould a sample solution that has a high absorbance reading, have a low or a high concentrationarrow_forward
- If water were not polar, would it dissolve sodium chloride? Explain.arrow_forwardIn not more than 10 sentences, discuss briefly why is water called the universal solvent?arrow_forwardAssume that you have a stock solution of 10% SDS and you need to make 150 microliters of 0.5% SDS. What volume of 10% SDS and what volume of water would you use?arrow_forward
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College

