
Concept explainers
(a)
Interpretation: The product formed by the treatment of given alcohol with
Concept introduction: The reaction of alcohols with halogen acids
Answer to Problem 9.39P
The products formed by the treatment of given alcohol with
Explanation of Solution
The given structure of alcohol is in the form of ball-and-stick model. It is converted into skeletal structure by replacing black ball with
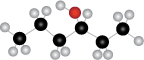
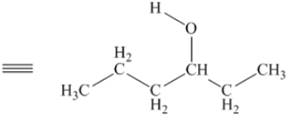
Figure 1
The above skeletal structure indicates that the given alcohol is secondary. It contains only one stereogenic center at
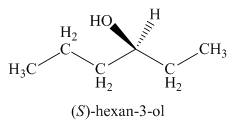
Figure 2
The reaction of alcohols with halogen acids
Since the given alcohol is secondary, it follows
The
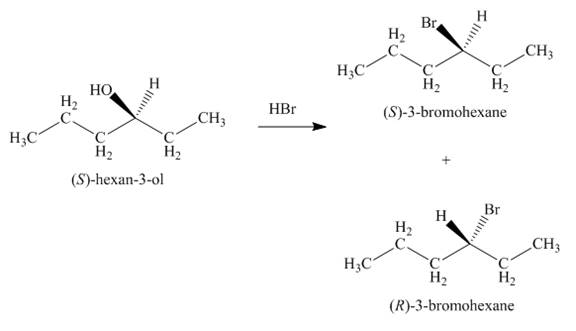
Figure 3
The products formed by the treatment of given alcohol with
(b)
Interpretation: The product formed by the treatment of given alcohol with
Concept introduction: Alkyl bromides are obtained by the reaction of
Answer to Problem 9.39P
The product formed by the treatment of given alcohol with
Explanation of Solution
Alkyl bromides are obtained by the reaction of
Since the given alcohol is secondary, it follows
The
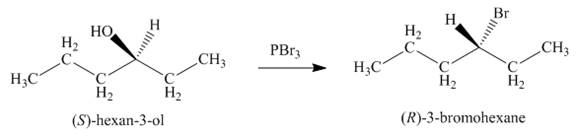
Figure 4
The product formed by the treatment of given alcohol with
(c)
Interpretation: The product formed by the treatment of given alcohol with
Concept introduction: The reaction of alcohols with halogen acids
Answer to Problem 9.39P
The products formed by the treatment of given alcohol with
Explanation of Solution
The reaction of alcohols with halogen acids
Since the given alcohol is secondary, it follows
The
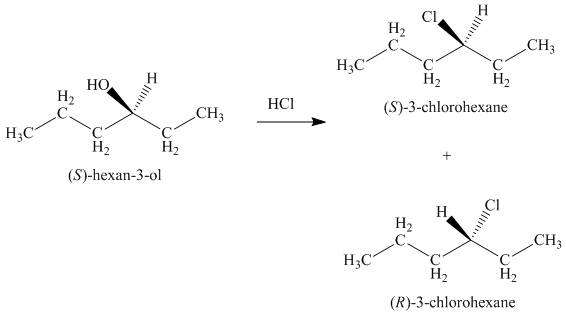
Figure 5
The products formed by the treatment of given alcohol with
(d)
Interpretation: The product formed by the treatment of given alcohol with
Concept introduction: Alkyl chlorides are obtained by the reaction of
Answer to Problem 9.39P
The product formed by the treatment of given alcohol with
Explanation of Solution
Alkyl chlorides are obtained by the reaction of
Since the given alcohol is secondary, it follows
The
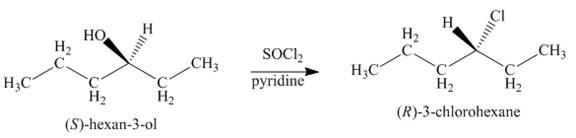
Figure 6
The product formed by the treatment of given alcohol with
Want to see more full solutions like this?
Chapter 9 Solutions
ORGANIC CHEMISTRY (LOOSELEAF) >CUSTOM<
- Draw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?arrow_forwardWhat happens when (i) chlorobenzene is treated with Cl2/FeCl3,(ii) ethyl chloride is treated with AgNO2, and(iii) 2-bromopentane is treated with alcoholic KOH?arrow_forwarda. What alkane, with molecular formula C5H12, forms only one monochlorinated product when it is heated with Cl2? b. What alkane, with molecular formula C7H16, forms seven monochlorinated products (disregarding stereoisomers) when heated with Cl2?arrow_forward
- Nitromethane is reacted with ethyl prop-2-enoate with EtO-Na+, EtOH 3 equivalents to give the product X(C16). X then reacts with H2/Raney Ni to give the product Y(C14), which in turn reacts with Na to give Z (C12). Indicate which products X, Y and Z are.arrow_forwardWhat happens when (i) Chlorobenzene is treated with Cl2/FeCl3 (ii) ethyl chloride is treated with AgN02, (iii) 2-bromopentane is treated with alcoholic KOH? Write the chemical equations in support of your answer.arrow_forwardDraw the organic products formed when cyclopentene is treated withfollowing reagent. [1] CH3CO3H; [2] H2O, HO−arrow_forward
- What alkane, with molecular formula C7H16, forms seven monochlorinated products (disregarding stereoisomers) when heated with Cl2?arrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. c. CH3CH2Cl, AlCl3 l. product in (c), then KMnO4arrow_forwardWhich alcohols can be prepared as a single product by hydroboration– oxidation of an alkene? Which alcohols can be prepared as a single product by the acid-catalyzed addition of H2O to an alkene?arrow_forward
- 1) The acid-catalyzed dehydration of 2-methyl-2-butanol yields two alkene products, what are the names of the two alkenes? 2) which of the two alkenes is the major product?arrow_forwardWhich alcohols can be prepared as a single product by hydroboration–oxidation of an alkene? Which alcohols can be prepared as a singleproduct by the acid-catalyzed addition of H2O to an alkene?arrow_forwardCH3-CH2-OH reacts with H+/140C = ? + H2Oarrow_forward
