
Concept explainers
(a)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane into
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by the other nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
The structure of the nucleophile that could be used to convert them into
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into the
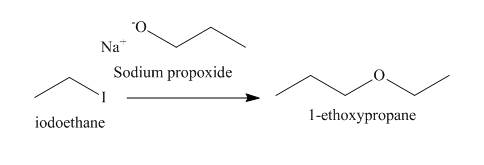
Figure 1
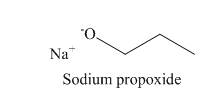
The structure of the nucleophile that could be used to convert the iodoethane to give

Figure 2
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown in Figure 2.
(b)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane to give the
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
The structure of the nucleophile that could be used to convert the iodoethane to give the
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into
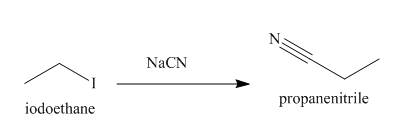
Figure 3
The structure of the nucleophile that could be used to convert the iodoethane to give the
The structure of the nucleophile that could be used to convert the iodoethane to give the
(c)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is to be stated.
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
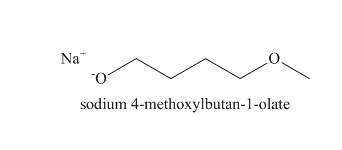
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown below.
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into the given compound is shown below.
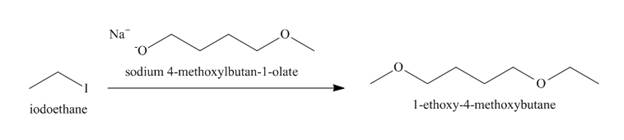
Figure 4
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown below.

Figure 5
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown in the Figure 5.
(d)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is to be stated.
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown below.
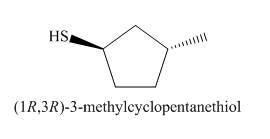
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into the given compound is shown below.
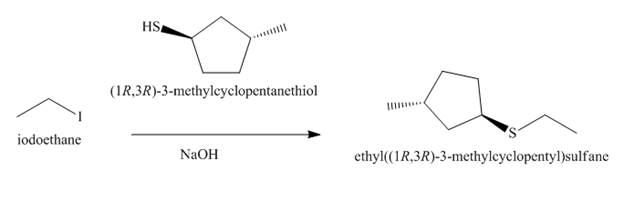
Figure 6
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown below.

Figure 7
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown in the Figure 7.
(e)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane to give
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
The structure of the nucleophile that could be used to convert the iodoethane to give
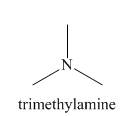
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into
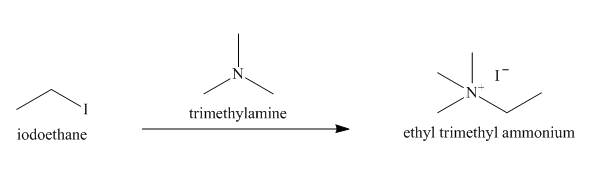
Figure 8
The structure of the nucleophile that could be used to convert the iodoethane to give

Figure 9
The structure of the nucleophile that could be used to convert the iodoethane to give
Want to see more full solutions like this?
Chapter 9 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- When A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With this information, write the correct structure of B and propose the reaction mechanism (step by step, with the correct use of arrows) to obtain B.arrow_forwardThree isomeric pentanols with unbranched carbon chains exist. Which of these isomers, upon dehydration at 180C, yields only 1-pentene as a product?arrow_forwardPlease be clear in your writing, solve step by step Compound A (C9H12), when hydrogenated by catalysis on Pd / C, absorbs 3 equivalents of H2 to give compound B (C9H18). Ozonolysis of compound A gives cyclohexanone (C6H10O). Compound A reacting with NaNH2 / NH3 followed by addition of CH3Br gave compound C (C10H14). What are the structures of compounds A, B, and C?arrow_forward
- Compound A is an alcohol that undergoes oxidation to produce compound B.Compound B is a ketone that gives positive triiodomethane reaction. Compound B isthen reacted with phenyl magnesium bromide, C6H5MgBr in the presence of aqueousacid to form compound C. Compound C has the molecular formula of C9H12O. Deducethe structure for compound A, B and C. PLEASE PROVIDE CLEAR DRAWINGS AND EXPLANATIONSarrow_forwardStarting with the following compounds, outline a practical synthesis of 1-butanolarrow_forwardStearolic acid, C18H32O2, yields stearic acid on catalytic hydrogenation and undergoes oxidative cleavage with ozone to yield nonanoic acid and nonanedioic acid. What is the structure of stearolic acid?arrow_forward
- When 5-bromo-1-pentanol is treated with sodium hydride in diethyl ether, the product is analyzed to be C5H10O. Propose a likely structure for this product, suggesting a reasonable mechanistic pathway for its formationarrow_forwardWrite the structure of the principal organic product formed in the reaction of 1-propanol with each of the following reagents:(a) Sulfuric acid (catalytic amount), heat at 140°C(b) Sulfuric acid (catalytic amount), heat at 200°C(c) Dimethyl sulfoxide (DMSO), oxalyl chloride [(COCl)2], triethylamine [N(CH2CH3)3](d) Pyridinium chlorochromate (PCC) in dichloromethane(e) Potassium dichromate (K2Cr2O7) in aqueous sulfuric acid, heat(f) Sodium amide (NaNH2)arrow_forwardWhen the compound shown below undergoes acid-catalyzed dehydration, a ring expansionoccurs to give the fused-ring product. What type of intermediate is formed in this reaction?Explain the ring expansion in terms of the reaction mechanismarrow_forward
- An organic compound A of unknown structure was found to have a molecular formula C8H16. When A was poured in water and heated, compound B having a molecular formula C8H18O was formed. B upon heating with sulfuric acid was converted to C as the major product which is identical to A. Ozonolysis of C gave one molecule each of two different products D and E, both having a molecular formula C4H8O. Write the reactions involved and determine the structure of A,B,C,D and E.arrow_forwardWrite the structure of the major organic product formed in the reaction of 1-pentene with each of the following: (a) Hydrogen chloride (b) Dilute sulfuric acid (c) Diborane in diglyme, followed by basic hydrogen peroxide (d) Bromine in carbon tetrachloride (e) Bromine in water (f) Peroxyacetic acid (g) Ozone (h) Product of part (g) treated with zinc and water (i) Product of part (g) treated with dimethyl sulfide (CH3)2Sarrow_forwardA hydrocarbon (X), with the molecular formula: C8H14 is reduced in presence of sodium and liquid ammonia to give the only product (Y) with the molecular formula: C8H16. Compounds X and Y both resulting 2,5-dimethylhexane when treated with hydrogen and platinum catalyst (H2/Pt). As a result of the oxidative cleavage of compound Y (by using KMnO4 / H2SO4), a single carboxylic acid derivative with C4H8O2 molecular formula is formed. Again, as a result of the reaction of Y with perbenzoic acid, the chiral compound C8H14O is observed, but the reaction of compound Y with bromine gives the achiral C8H14Br2 as the product.arrow_forward

 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



