
Concept explainers
(a)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane into
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by the other nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
The structure of the nucleophile that could be used to convert them into
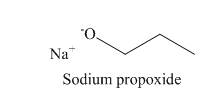
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into the
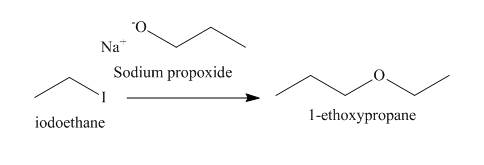
Figure 1
The structure of the nucleophile that could be used to convert the iodoethane to give

Figure 2
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown in Figure 2.
(b)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane to give the
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
The structure of the nucleophile that could be used to convert the iodoethane to give the
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into
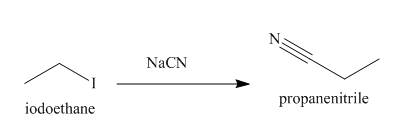
Figure 3
The structure of the nucleophile that could be used to convert the iodoethane to give the
The structure of the nucleophile that could be used to convert the iodoethane to give the
(c)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is to be stated.
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown below.
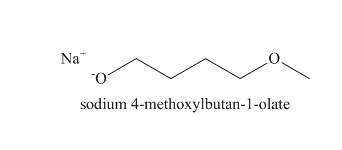
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into the given compound is shown below.
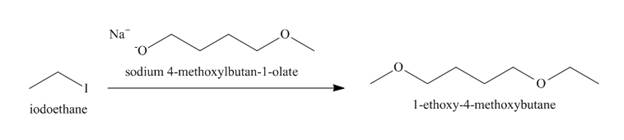
Figure 4
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown below.

Figure 5
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown in the Figure 5.
(d)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is to be stated.
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown below.
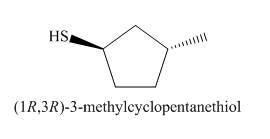
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into the given compound is shown below.
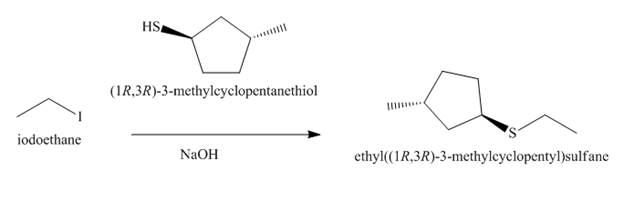
Figure 6
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown below.

Figure 7
The structure of the nucleophile that could be used to convert the iodoethane to give the given compound is shown in the Figure 7.
(e)
Interpretation:
The structure of the nucleophile that could be used to convert the iodoethane to give
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the incoming nucleophile.
The
Answer to Problem 9.52AP
The structure of the nucleophile that could be used to convert the iodoethane to give
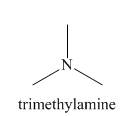
Explanation of Solution
The
The reaction that takes place to convert the iodoethane into
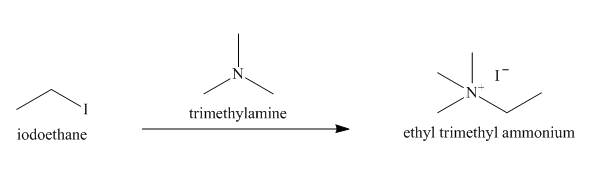
Figure 8
The structure of the nucleophile that could be used to convert the iodoethane to give

Figure 9
The structure of the nucleophile that could be used to convert the iodoethane to give
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry Study Guide and Solutions
- Predict the major products formed when benzoyl chloride (PhCOCl) reacts with the following reagents.(a) ethanol (b) sodium acetate (c) anilinearrow_forwardPredict the major products formed when benzoyl chloride (PhCOCl) reacts with the following reagents.(a) ethanoarrow_forward(b) State the reagents needed to convert benzoic acid into the following compounds. (i) C6H§COCI (ii) C,H$CH2OH (iii) C6H$CONHCH3arrow_forward
- Predict the major product or the necessary reagent or reactant to complete each of the following reactions. In the box before each reaction, indicate the mechanism followed by the reaction. (Free radical (FR), SN2, SN1). (b) (a) Cl₂ CH₂CH₂OH (a) 1 hv 2 (b) H Br H₂O Na+ SCH3 (c) (d) CI "CH3arrow_forward(a) How will you convert:(i) Benzene to acetophenone (ii) Propanone to 2-Methylpropan-2-ol(b) Give reasons :(i) Electrophilic substitution in benzoic acid takes place at meta position.(ii) Carboxylic acids are higher boiling liquids than aldehydes, ketones and alcohols of comparable molecular masses.(iii) Propanal is more reactive than propanone in nucleophilic addition reactions.arrow_forwardGive reasons for the following: (i) p-nitrophenol is more acidic than p-methylphenol. (ii) Bond length of C—O bond in phenol is shorter than that in methanol. (iii) (CH3)3C—Br on reaction with sodium methoxide (Na+ _OCH3) gives alkene as the main product and not an ether.arrow_forward
- Draw line structures of the following compounds and the product you would obtain from the reduction of each.(a) Isopropyl methyl ketone (b) p-Hydroxybenzaldehyde(c) 2-Methylcyclopentanonearrow_forward(a) rate at which the following alcohols undergo dehydration to form alkenes in sulfuric acid OH OH OH (b) rate at which the following alkenes undergo addition with HBr to form an alkyl halidearrow_forwardDraw the structural formula for the major product of the following reactions.arrow_forward
- Which is the major isolated organic product of the reaction shown? (A) (B) Pd(PPH3)4 KOHarrow_forwardGive reasons for the following :(i) Phenol is more acidic than methanol.(ii) The C—O—H bond angle in alcohols is slightly less than the tetrahedral angle (190°28′).(iii) (CH3)3C—O—CH3 on reaction with HI gives (CH3)3C—I and CH3—OH as the main products and not (CH3)3C—OH and CH3—I.arrow_forward(a) What functional group is undergoing a transformation in the reaction? (b) What functional group is it being transformed into (in the final product)?arrow_forward
