General, Organic, and Biological Chemistry Plus Mastering Chemistry with Pearson eText - Access Card Package (3rd Edition)
3rd Edition
ISBN: 9780134041568
Author: Laura D. Frost, S. Todd Deal
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 9.82AP
Consider the lactic-acid buffer with lactic acid, CH3CH(OH)COOH and its salt sodium lactate, NaCH3CH(OH)COO:
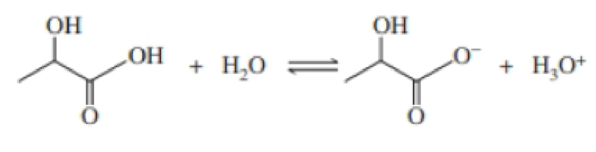
- a. What is the purpose of a buffer system?
- b. Why is a salt of the acid needed?
- c. How does the buffer react when acid (H3O+) is added?
- d. How does the buffer react when base (OH−) is added?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The muscle pain and fatigue felt during strenuous exercise is caused by the buildup of lactic acid (HC3H5O3) in muscle tissues. In a 1.00M solution of lactic acid, the acid is 2.94% ionized.
a) Write the equation for dissociation.
b) Write the Ka expression for the acid.
c) What is the equilibrium concentration of each product and the reactant?
d) What is the value of Ka?
e) What is the pH of the solution?
Methylamine is a weak base with K, = 4.38 x 104. 25.0 mL of a 0.50 M solution of methylamine is reacted with a solution of 0.20 M HCI
. A) What is the pH after a total of 62.5 mL of HCl is added.
B) What is the pH after a total of 75.0 mL of HCI is added
a. Calculate the [OH⁻] of baking soda with [H₃O⁺]=1.8 x 10⁻⁸ M b. Calculate the [OH⁻] of blood with [H₃O⁺]=4.2 x 10⁻⁸ M
Chapter 9 Solutions
General, Organic, and Biological Chemistry Plus Mastering Chemistry with Pearson eText - Access Card Package (3rd Edition)
Ch. 9 - Prob. 9.1PPCh. 9 - Indicate if each of the following statements is...Ch. 9 - Prob. 9.3PPCh. 9 - Prob. 9.4PPCh. 9 - Prob. 9.5PPCh. 9 - In each of the following equations, identify the...Ch. 9 - Prob. 9.7PPCh. 9 - Prob. 9.8PPCh. 9 - Prob. 9.9PPCh. 9 - Which of the following are strong bases? a....
Ch. 9 - Prob. 9.11PPCh. 9 - Prob. 9.12PPCh. 9 - Prob. 9.13PPCh. 9 - Prob. 9.14PPCh. 9 - Complete and balance the following neutralization...Ch. 9 - Complete and balance the following neutralization...Ch. 9 - Prob. 9.17PPCh. 9 - Complete and balance the following neutralization...Ch. 9 - Prob. 9.19PPCh. 9 - Prob. 9.20PPCh. 9 - Write an equilibrium constant expression lor the...Ch. 9 - Prob. 9.22PPCh. 9 - Prob. 9.23PPCh. 9 - Prob. 9.24PPCh. 9 - Prob. 9.25PPCh. 9 - Sulfur trioxide is produced by reacting sulfur...Ch. 9 - Prob. 9.27PPCh. 9 - Prob. 9.28PPCh. 9 - Prob. 9.29PPCh. 9 - When you exercise, energy is produced by...Ch. 9 - Using Tables 9.1 and 9.6, identity the stronger...Ch. 9 - Using Tables 9.1 and 9.6, identify the stronger...Ch. 9 - Prob. 9.33PPCh. 9 - Identify the acid and base on the reactant side of...Ch. 9 - Prob. 9.35PPCh. 9 - Prob. 9.36PPCh. 9 - Prob. 9.37PPCh. 9 - Write the formula and name of the conjugate acid...Ch. 9 - Complete the following reactions and identify the...Ch. 9 - Complete the following reactions and identify the...Ch. 9 - State if each of the following solutions is...Ch. 9 - State if each of the following solutions is...Ch. 9 - State if each of these following solutions is...Ch. 9 - Slate if each of the following solutions is...Ch. 9 - Calculate the pH of each of the solutions in...Ch. 9 - Calculate the pH of each of the solutions in...Ch. 9 - Calculate the [H3O+] for each of the following...Ch. 9 - Calculate the [H3O+J lor each of the following...Ch. 9 - Prob. 9.49PPCh. 9 - Using Table 9.8, determine the stronger acid from...Ch. 9 - Prob. 9.51PPCh. 9 - Prob. 9.52PPCh. 9 - Prob. 9.53PPCh. 9 - Prob. 9.54PPCh. 9 - Valine has the zwitterion structure shown in the...Ch. 9 - Glycine has the zwitterion structure shown in the...Ch. 9 - Prob. 9.57PPCh. 9 - Prob. 9.58PPCh. 9 - During stress or trauma, a person can start to...Ch. 9 - A person who overdoses on antacids may neutralize...Ch. 9 - Prob. 9.61APCh. 9 - Prob. 9.62APCh. 9 - Prob. 9.63APCh. 9 - What are some ingredients found in antacids? What...Ch. 9 - Prob. 9.65APCh. 9 - Prob. 9.66APCh. 9 - Prob. 9.67APCh. 9 - Prob. 9.68APCh. 9 - For the following reaction, 2HI(g)H2(g)+I2(g) a....Ch. 9 - Prob. 9.70APCh. 9 - Prob. 9.71APCh. 9 - Prob. 9.72APCh. 9 - Prob. 9.73APCh. 9 - Prob. 9.74APCh. 9 - Prob. 9.75APCh. 9 - Prob. 9.76APCh. 9 - Determine the pH for the following solutions....Ch. 9 - Determine the pi! for the following solutions....Ch. 9 - Prob. 9.79APCh. 9 - Prob. 9.80APCh. 9 - Consider the acetic-acid buffer system with acetic...Ch. 9 - Consider the lactic-acid buffer with lactic acid,...Ch. 9 - In blood plasma, pH is maintained by the carbonic...Ch. 9 - Adding a few drops o! a strong add to water will...Ch. 9 - Consider the amino acid valine shown in its...Ch. 9 - Prob. 9.86CPCh. 9 - Prob. 9.87CPCh. 9 - Prob. 9.88CPCh. 9 - Prob. 9.89CPCh. 9 - Naproxen, the active ingredient in Aleve has the...Ch. 9 - To determine the concentration of an unknown weak...Ch. 9 - Explain why the following amino acid cannot exist...Ch. 9 - Prob. 1IA.1QCh. 9 - Prob. 1IA.2QCh. 9 - Prob. 1IA.3QCh. 9 - Prob. 1IA.4QCh. 9 - Prob. 1IA.5QCh. 9 - Provide the a. conjugate base of H2S. _______ b....Ch. 9 - If the lungs fail to expel normal amounts of CO2...Ch. 9 - If the lungs expel CO2 faster than normally...Ch. 9 - Prob. 2IA.3QCh. 9 - Prob. 2IA.4QCh. 9 - Prob. 1ICCh. 9 - Prob. 2ICCh. 9 - Prob. 3IC
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the pH of 1.000 L of a solution of 100.0 g of glutamic acid (C5H9NO4, a diprotic acid; K1=8.5105,K2=3.391010) to which has been added 20.0 g of NaOH during the preparation of monosodium glutamate, the ?avoring agent? What is the pH when exactly 1 mol of NaOH per mole of acid has been added?arrow_forwardExplain why a buffer can be prepared from a mixture of NH4Cl and NaOH but not from NH3 and NaOH.arrow_forwardA) how many miles of NaOH were required to reach the equivalence point ? B) how many moles of NaOH were required to reach the equivalence point ? C) How many moles of CH3COOH were present in the initial sample of acid ? D) what was the exact concentration of the initial acetic solution ?arrow_forward
- pH Calculations of Polyprotic Systems b.)What is the pH of 0.1000 M NaH2PO4? (Ka1 = 6.9x10-3, Ka2=6.2x10-8) c.)What is the pH of 0.1000 M Na2HPO4? (Ka2=6.2x10-8, Ka3=4.8x10-13) d.)What is the pH of 0.1000 M Na3PO4? (Ka3=4.8x10-13)arrow_forwardHow do buffers work? Explain using H2CO3-NaHCO3 buffer by writing the equations for: (a) dissociation with water (b) addition of HCl (c) addition of NaOHarrow_forwardHw.99. What is the resulting pH of a solution that forms when 15.00 mL of a 0.150 mol/L hydrochloric acid solution combined with 10.00 mL of a 0.150 mol/L sodium hydroxide solution?arrow_forward
- A phosphate buffer contains KH2PO4 and K2HPO4. What is the net ionic equation for the buffering reaction that occurs when some HCl is added to the buffer solution? (A) H2PO4–(aq) + Cl–(aq) --> HPO42–(aq) + HCl(aq); (B) H2PO4–(aq) + H3O+(aq) --> H3PO4(aq) + H2O(aq); (C) HPO42–(aq) + Cl–(aq) --> PO43–(aq) + HCl(aq); (D) HPO42–(aq) + H3O+(aq) --> H2PO4–(aq) + H2O(aq);arrow_forwardA 0.018 M solution of salicylic acid, HOC6H4CO2H, has the same pH as 0.0038 M HNO3solution. (a) Write an equation for the ionization of salicylic acid in aqueous solution. (Assume only the –CO2H portion of the molecule ionizes.) (b) What is the pH of solution containing 0.018 M salicylic acid? (c) Calculate the Ka of salicylic acid.arrow_forwardA sample of saliva has a pH of 5.8 Which statement describing the saliva sample is NOT true? (a) The saliva sample has a [OH- ] greater than its [H3O+ ]. (b) The saliva sample is acidic. (c) The saliva sample has a [H3O+ ] concentration greater than 1.0 x 10-7 M (d) The saliva sample has a [H3O+ ] concentration than that of a neutral solution.arrow_forward
- CH3CH2NH2 is a weak base, and HCl is a strong acid. One mole of the salt, CH3CH2NH3+Cl–, is added to one liter of wate. Will the solution be acidic, basic, or neutral? Which reaction predominates in the solution described in this question? Cl– + H2O <--> HCl + OH- CH3CH2NH3+ + H2O <--> CH3CH2NH2 + H3O+ H2O + H2O <--> H3O+ + OH- CH3CH2NH3+ + H2O <--> CH3CH2NH42+ + OH- - The Kb for CH3CH2NH2 is 6.4x10-4. What is the pH of the solution described in this question?arrow_forwardWhat is the [H3O+] of a solution that is pH 4.2? The conjugate base of a reactant in the following equation is H2SO4 + NH3 ⇆ NH4 + + HSO4 – What happens when NaOH is added to a buffer solution of HF/NaF? Which of the following would constitute a buffer solution? a. H2CO3/NaHCO3 b. HCl/NaOH c. NaHSO4/ H2SO4 d. H2CO3/HCl What is the pH of a 0.0035 M solution of NaOH? Which of the following is the weakest acid? a. HOCl (Ka = 3.0 X 10-8) b. CH3COOH (Ka = 1.8 X 10-5) c. HF (Ka = 6.8 X 10-4) d. HCN (Ka = 6.2 X 10-10)arrow_forwardEthylene glycol (antifreeze) is converted to a(n) __________ in the liver. inorganic acid base amine carboxylic acid toxic gas A buffer works best when the concentration of weak acid is high and ___________ its conjugate base's concentration. equal to twice 1/10 of 1/100 of 100Xarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY