
Interpretation:
The required volume of stock solution to prepare the given volumes and concentrated final solutions are should be calculated by using given representation diagram.
Concept introduction:
Dilution:
- Dilution is the process, which is used to prepare a low concentrated solution from high concentrated solution.
Volumetric principle:
- In the dilution process, the relationship between initial and final concentrations and volumes of solutions are given in the volumetric equation and it is,
Explanation of Solution
To calculate the required volumes of stock solutions to prepare the final diluted solutions
The given diagram is,
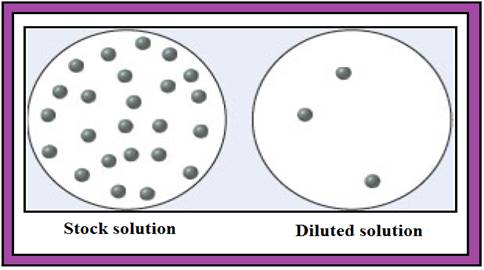
Diagram.1
To calculate the required volume of stock solution to prepare
From the given diagram (Diagram.1) the stock solution contains 24 numbers of solute particles and diluted solution contains 3 numbers of solute particles.
Consider a unit of stock solution contains 24 numbers of solute particles and a unit of diluted solution contains 3 numbers of solute particles.
The numbers of solute particles and final volume are plugged in the above equation to give required volume of stock solution to prepare
The required volume of stock solution to prepare
The required volume of stock solution to prepare
From the given diagram (Diagram.1) the stock solution contains 24 numbers of solute particles and diluted solution contains 3 numbers of solute particles.
Consider a unit of stock solution contains 24 numbers of solute particles and a unit of diluted solution contains 3 numbers of solute particles.
The numbers of solute particles and final volume are plugged in the above equation to give required volume of stock solution to prepare
The required volume of stock solution to prepare
The required volume of stock solution to prepare
From the given diagram (Diagram.1) the stock solution contains 24 numbers of solute particles and diluted solution contains 3 numbers of solute particles.
Consider a unit of stock solution contains 24 numbers of solute particles and a unit of diluted solution contains 3 numbers of solute particles.
The numbers of solute particles and final volume are plugged in the above equation to give required volume of stock solution to prepare
The required volume of stock solution to prepare
The required volume of stock solution to prepare the given volumes and concentrated final solutions are calculated by using given representation diagram.
Want to see more full solutions like this?
Chapter 9 Solutions
GEN COMBO CHEMISTRY: ATOMS FIRST; ALEKS 360 2S ACCESS CARD CHEMISTRY:ATOMS FIRST
- What kinds of solute particles are present in a solution of an ionic compound? Of a molecular compound?arrow_forwardAn experiment in a general chemistry laboratory calls for a 2.00-M solution of HCl. How many mL of 11.9 M HCl would be required to make 250 mL of 2.00 M HCl?arrow_forwardRubbing alcohol contains 585 g isopropanol (C3H7OH) per liter (aqueous solution). Calculate the molarity.arrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





