
Practice calculating dilutions using the following problems.
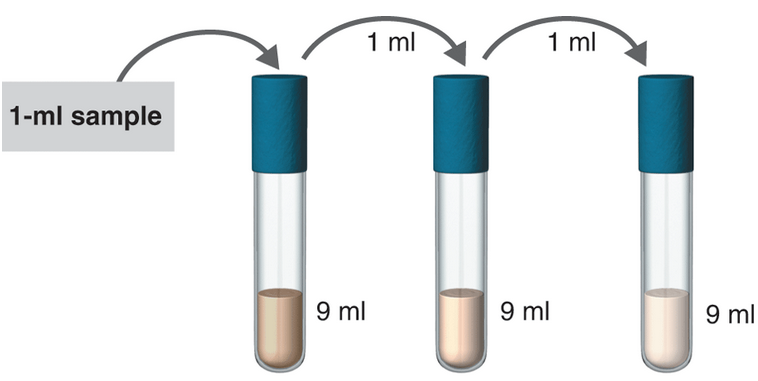
- Dilution:
To determine:
The serial dilution in the given samples.
Introduction:
The stepwise dilution of a solution is termed as a serial dilution. Generally, the dilution factor at each stage of serial dilution is constant and leads to geometric progression in a logarithmic manner of the concentration of the sample. Serial dilutions are used to dilute the given substance at a very high level and serial dilutions are also used in the formation of concentration curves.
Explanation of Solution
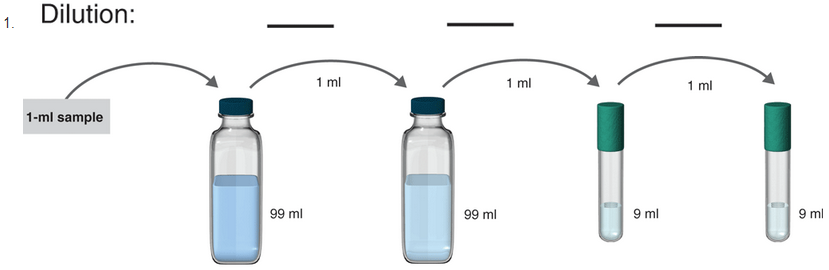
The samples where serial dilution is occurring is shown below:
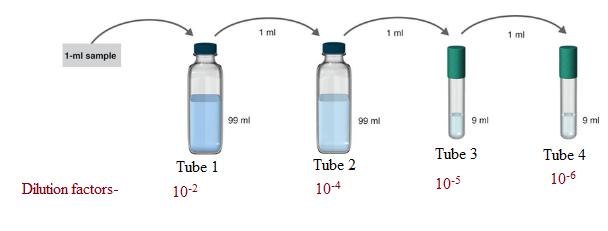
1 ml of the sample is mixed in 99ml solvent. So, the total solution is of 100 ml.
Hence, in tube 1 the serial dilution is of 1:100.
Now, 1 ml of the sample from tube 1 is poured to 99 ml solvent in tube 2. So, the total volume of the solution in tube 2 becomes 100 ml.
Hence, in tube 2 the serial dilution is now =
Now, 1 ml of the sample from tube 2 is poured to 9 ml solvent in tube 3. So, the total volume of the solution in tube 3 becomes 10 ml.
Hence, in tube 3 the serial dilution is now =
Now, 1 ml of the sample from tube 3 is poured to 9 ml solvent in tube 4. So, the total volume of the solution in tube 4 becomes 10 ml.
Hence, in tube 4 the serial dilution is now =
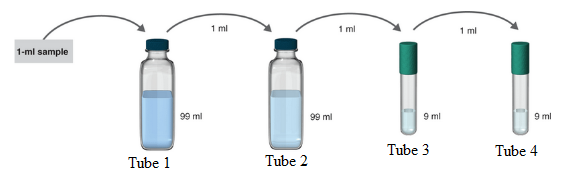
The samples where serial dilution is occurring is shown below:
Want to see more full solutions like this?
Chapter B Solutions
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
- When determining CFUs for a dilution of the same sample at 10-2 and 10-3 which of the following results would seem the most accurate to you? Explain why in the space below. A. 110,000 CFU/mL for 10-2; and 140,000 CFU/mL 10-3 B. 110,000 CFU/mL for 10-2; and 14,000 CFU/mL 10-3 C. 10,000 CFU/mL for 10-2; and 140,000 CFU/mL 10-3 D. none of the abovearrow_forwardSketch a potentiometric biosensor measurementarrow_forwardwhat are the buffers used for biological and clinical measurements? state the properties of each buffer and the limitation to its applications.arrow_forward
- give the respective the TOTAL DILUTION FACTOR of respective serial dilutions. show complete solution.arrow_forwardHow would you perform a 100-fold dilution, if you want the final volume to be 15mL? What volume of sample and volume of dilute would be used?arrow_forwardUsing the fraction conversion method solve the following Convert 500 mL=______qtarrow_forward
- During transfer of liquids using a micropipette: What would it mean if the mean is very close to the expected value but your standard deviation is high ?arrow_forwardLabel the void volume (V0), elution volume (Ve) and total elution volume (Vt) appropriately on the graph for the gel filtration chromatography using a column experiment.arrow_forwardShow workflow process of producing a Liquid Dosage Form through Extemporaneous CompoundingUse diagrams (boxes and arrows) to signify stepwise and significant procedures.arrow_forward
- Make a list of the five most critical qualities of an effective extraction buffer. Please provide a brief description of each.arrow_forward1. compute for patient B result of his chloride analysis with the following data using mg/dL unit factor: 355/ mg/dL , Absorbance sample = 0.2148, Absorbance standard = 0.3017 a. 24.8 mg/L b. 25.27 mg/dL c. 2.7119 mmol/L d. 252.7 mg/dL 2. an adult patient suffering from debilitation is supected of calcium deficiency. His doctor requested for calcium anaylusis with the following data gathered standard known value is B: Absorabance is 0.2876. sample absorbance is 0.4291 compute for the calcium in mmol/L a. 53.6mmol/L b. 5.36 mmol/L c. 11.9 mmol/L d. 119mmol/L 3. calculate the value of sodium where the PREC solution has diclored. data gathered are the following: Absorbance of Reagent Blank = 0.3124 Absorbance of sample = 0.114, Absorbance of standard = 0.275 value is 150mmol/L a. 108.2 mmol/L b. 170.4 mmol/L c. 795.72 mmol/L d. 79.5 mmol/Larrow_forwardIllustrate the dilution series used and label the final dilution of each dilution.arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning



