
Concept explainers
Bromination of 2-methylbutane yields predominantly one product with the formula
(a) 2-Methyl-2-butene
(b) 2-Methyl-2-butanol
(c) 3-Methyl-2-butanol
(d) 3-Methyl-1-butyne
(e) 1-Bromo-3-methylbutane
(f) 2-Chloro-3-methylbutane
(g) 2-Chloro-2-methylbutane
(h) 1-Iodo-3-methylbutane
(i)
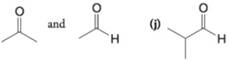
Want to see the full answer?
Check out a sample textbook solution
Chapter FRP Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Additional Science Textbook Solutions
Introductory Chemistry (6th Edition)
Chemistry: Structure and Properties (2nd Edition)
Living By Chemistry: First Edition Textbook
Chemistry: A Molecular Approach
- The following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forward(a) Why is the following reaction a poor method for synthesizing tert-butyl propyl ether? (b) What would be the major product of this reaction? (c) Propose a better synthesis of tert-butyl propyl ether. does not give CH3 CH,CH,CH,—O: Na + CH;–c–Br CH3 tert-butyl bromide odium propoxide CH3 CH₂-C-0-CH₂CH₂CH T CH3 tert-butyl propyl etherarrow_forward
- (a) Why is the following reaction a poor method for the synthesis of t-butyl propyl ether? (b) What would be the major product from this reaction? (c) Propose a better synthesis of r-butyl propyl ether. ÇH, does not give ÇH, CH,CH,CH,-O: *Na + CH,-C-Br > CH,-C-o-CH,CH,CH, CH, CH, t-butyl propyl ether sodium propoxide 1-butyl bromidearrow_forwardHow could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1-Butanol (b) 1-Bromobutane (c) Pentanoic acid (d) 1-Butene (e) Octanearrow_forward4. Synthesize the following compound. You may use benzene, acetylene, ethanol and any inorganic reagents. O₂Narrow_forward
- Explain how you would synthesize the following compounds. As raw materials you can use any alcohols containing four or fewer carbon atoms, cyclohexanol, and any necessary inorganic solvents and reagents.arrow_forward1. Predict the elimination products of the following reactions. When two alkenes are possible, predict which one will be the major product. Explain your answers, showing the degree of substitution of each double bond in the products. 2. Which of these reactions are likely to produce both elimination and substitution products? (a) 2-bromopentane +NaOCH3 (b) 3-bromo-3-methylpentane +NaOMe(Me= methyl, CH3) (c) 2-bromo-3-ethylpentane +NaOH (d) cis-1-bromo-2-methylcyclohexane +NaOEt (Et= ethyl, CH2CH3)arrow_forward1. How would you prepare the following alkyl halides from the appropriate alcohols? (a) ÇI (b) Br CH3 CH3CH3 CH;CHCH,CHCH3 ČH3 2. Predict the products of the following reaction: (a) он CH3 (b) H2C CH;CH,CHCH,CHCH, Per, socl2. PBr3 ? H3Carrow_forward
- Use a Grignard reaction to prepare the following alcohols: (a) 2-Methyl-2-propanol (b) 1-Methylcyclohexanol (c) 3-Methyl-3-pentanol (d) 2-Phenyl-2-butanol (e) Benzyl alcohol (f) 4-Methyl-1-pentanolarrow_forwardb) Refer to the following equation to answer Q3b (i), (ii) and (iii). CH3 H,SO, Н—с—он C-CH3 ? + H2O Но- ČH3 (i) Determine the product of the above reaction. (ii) Name the above reaction. (iii) Propose the mechanism for the above reaction.arrow_forwardPredict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the stronger acid, the stronger base, the weaker acid, and the weaker base: (a) CH3CH=CH2 + NANH2 (d) CH3C=C: + CH;CH2OH → (e) CH3C=C:- + NH¾CI – | (b) CH;C=CH + NaNH2 (c) CH3CH2CH3 + NANH2 → | HASarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

