
EBK ORGANIC CHEMISTRY
12th Edition
ISBN: 9781119233664
Author: Snyder
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter H, Problem 6PP
Practice Problem H.6
Papaverine has been synthesized by the following route:
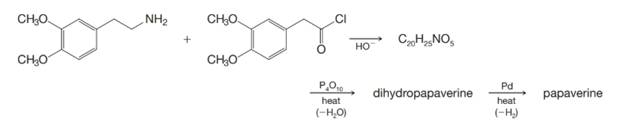
Outline the reactions involved.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Select all possible products of the following question
are the reactants correct to produce N-Ethyl-2-phenylethanamine and show mechanism
What compound undergoes metathesis to form each of the following compounds?
Chapter H Solutions
EBK ORGANIC CHEMISTRY
Ch. H - Prob. 1PPCh. H - Prob. 2PPCh. H - Prob. 3PPCh. H - Practice Problem H.4
In 1891 G. Merling...Ch. H - Practice Problem H.5
Many alkaloids appear to be...Ch. H - Practice Problem H.6 Papaverine has been...Ch. H - Practice Problem H.7
One of the important steps in...Ch. H - Prob. 8PPCh. H - Practice Problem H.9 Gramine has been synthesized...
Additional Science Textbook Solutions
Find more solutions based on key concepts
For each of the following, (i) give the systematic name of the compound and specify the oxidation state of the ...
General Chemistry: Atoms First
A circuit board with a dense distribution of integrated circuits (ICs) and dimensions of 120mm120mm on a side i...
Fundamentals of Heat and Mass Transfer
39. Consider the reaction:
Kp = 28.4 at 298 K
In a reaction mixture at equilibrium, the partial pressure...
Chemistry: Structure and Properties
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
a. Draw the resonance forms for SO2 (bonded OSO). b. Draw the resonance forms for ozone (bonded OOO). c. Sulfur...
Organic Chemistry (9th Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- PROBLEM 19.9 Predict the product and show the mechanism for the following reactions: (a) (b) excess NaOH excess I₂ excess NaOH excess I₂arrow_forwardOChem Question: Starting with propyne and using any other required reagents, show how you would synthesize (2R, 3R)- and (2S, 3S)-2,3-dibromobutane (as a racemic form)arrow_forwardProblem 15.3b Homework Predict the major organic products of the following reaction and draw their structures in the space provided. If appropriate, assume an aqueous workup at the end of the reaction. O HO + NaOMe 7 Open in Reading Viarrow_forward
- Identify the compounds below that cannot be made via a direct Friedel-Crafts alkylation but can be made via acylation followed by a Clemmensen reduction (to avoid carbocation rearrangements). Select all that apply. A) on B) D) O A В C D C C C Carrow_forwardWhich of the following synthesis reactions of organohalide occurs via electrophilic addition? (A) reaction of chlorine with propane B reaction of chlorine with propene reaction of thionyl chloride with propan-2-ol (D) all choicesarrow_forwardProblem 9.13 Write the mechanism of the acid-catalyzed reaction of methanol with cyclo- hexanone to give a hemiacetal.arrow_forward
- shift fn Onino dantbuogmo binspiCKey Reactions 549 Problem 14.21 (a) Draw the disulfide formed when CH,CH,CH,SH is oxidized. (b) Draw the product formed when the following disulfide is reduced: CH,CH,CH,CH,SSCH,CH,.arrow_forwardWhich of the following are a major product of the reaction sequence shown belo (1) Br2, FeBr3 (2) Mg/ether он он (A) (B) (3) (4) dil H* /H;O (C) (D) он Compound C Compounds B and D OCompounds A and B Compound Darrow_forward18.35 Draw the most likely mechanism for each of the following transformations: Br OH Br Br2 FeBr, 1) NaOH, 350°c 2) H,0* (a) (b) O2N O2N. 1) NaNH, 2) H,O" (c) NH2arrow_forward
- Problem 17.9: Predict the major products in each of the following. Please show the complete CO CN mechanisms, Thanks 1. LIAIH,, ether 2. Но (a) H,C. 1. LIAIH, ether 2. H,00 (b) 10arrow_forwardProblem 3.3 Draw mechanisms for each of the following substitution reactions. Remember that in each case, loss of the leaving group is preceded by coordination of the leaving group to a Lewis acid such as H+. (a) (b) CH3 CH3 HCl H3C- OH H3C- Cl CH3 CH3 OH ZnCl2 Ar Ar Ar Ar HOarrow_forward21.10 Predict the major product for each of the following transformations: (a) ملی OH 1) Br₂, PBr3 2) HO (b) .OH 1) Bra, PBr3 2) HO ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License