
Organic Chemistry, 12e Study Guide/Student Solutions Manual
12th Edition
ISBN: 9781119077329
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter SRP, Problem 27P
Interpretation Introduction
Interpretation:
The structure of the intermediate compound from A to M is to be written and the intermediate formed by the reaction of compound D with N-Bromo succinimide, the stereoisomer of Dianeackerone, are to be represented.
Concept introduction:
Dianeackerone consists of nineteen atoms of carbon, thirty atoms of hydrogen and one tom of oxygen due to which its molecular formula is 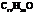 . The IUPAC name of this compound is
. The IUPAC name of this compound is 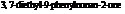 . It is a natural product and has a volatile behavior. It has both
. It is a natural product and has a volatile behavior. It has both 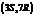 and
and 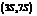 stereoisomers.
stereoisomers.
N-Bromo succinimide, also known as NBS, is a type of brominating agent, which is used as a source of bromine in many reactions.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)
Treatment of compound E (molecular formula C4H8O2) with excessCH3CH2MgBr yields compound F (molecular formula C6H14O) afterprotonation with H2O. E shows a strong absorption in its IR spectrum at1743 cm−1. F shows a strong IR absorption at 3600−3200 cm−1. The 1HNMR spectral data of E and F are given. What are the structures of E andF?Compound E signals at 1.2 (triplet, 3 H), 2.0 (singlet, 3 H), and 4.1 (quartet, 2 H) ppm Compound F signals at 0.9 (triplet, 6 H), 1.1 (singlet, 3 H), 1.5 (quartet, 4H), and 1.55 (singlet, 1 H) ppm
Compound C has the molecular formula C5H8O. The IR, 1H, 13C, and DEPT NMR spectra of this compound are shown below.
(a) Calculate the double bond equivalent of compound C and briefly explain what the values obtains represents. Interpret the IR spectrum.
(b) Based on the information provided, determine the structure of compound D.
Chapter SRP Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Ch. SRP - 1. Arrange the compounds of each of the following...Ch. SRP - 2. Arrange the compounds of each of the following...Ch. SRP - Predict the final product from each of the...Ch. SRP - Prob. 4PCh. SRP - Write detailed mechanisms for each of the...Ch. SRP - Prob. 6PCh. SRP - Prob. 7PCh. SRP - Prob. 8PCh. SRP - Prob. 9PCh. SRP - Give stereochemical structures for compounds AD:
Ch. SRP - Prob. 11PCh. SRP - The remaining steps in the industrial synthesis of...Ch. SRP - Prob. 13PCh. SRP - Prob. 14PCh. SRP - Prob. 15PCh. SRP - Prob. 16PCh. SRP - 17. Show how you would modify the synthesis given...Ch. SRP - Prob. 18PCh. SRP - Give structures for compounds AD. Compound D gives...Ch. SRP - The tranquilizing drug meprobamate (Equanil or...Ch. SRP - Prob. 21PCh. SRP - 22. Outlined here is the synthesis of a central...Ch. SRP - 23. What are compounds A and B? Compound B has a...Ch. SRP - Prob. 24PCh. SRP - 25. The Dow process for synthesizing phenol, which...Ch. SRP - Prob. 26PCh. SRP - Prob. 27PCh. SRP - 28. Compound Y shows prominent IR absorption...Ch. SRP - Prob. 29PCh. SRP - Consider this reaction involving peracetic acid:...Ch. SRP - 31. A compound (N) with the molecular formula...Ch. SRP - 32. Compound X is insoluble in aqueous sodium...Ch. SRP - Write the structures of the three products...Ch. SRP - Compound C (C9H11NO) gives a positive Tollens test...Ch. SRP - A compound X (C10H14O) dissolves in aqueous sodium...Ch. SRP - Compound Z (C5H10O) decolorizes bromine. The IR...Ch. SRP - 37. Compound W was isolated from a marine annelid...Ch. SRP - 38. Phenols generally are not changed on treatment...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Treatment of 1,3,6-cyclononatriene (Compound 1), or its dimethyl derivative (Compound 2), with potassium amide (KNH₂) in liquid ammonia results in the formation of anion 1a or 2a, respectively (J. Am. Chem. Soc. 1973, 95, 3437-3438): 9 15.85a 2 6 3 4 5 Compound 1 (R=H) Compound 2 (R=CH3) * You answer is incorrect. KNH₂ KNH₂ 1a (anion) 2a (anion) Of the following, which is NOT one of the four resonance structures of 1a?arrow_forwardAs reaction of (CH3)2CO with LIC≡CH followed by H2O affords compound D, which has a molecular ion in its mass spectrum at 84 and prominent absorptions in its IR spectrum at 3600−3200, 3303, 2938, and 2120 cm−1. D shows the following 1H NMR spectral data: 1.53 (singlet, 6 H), 2.37 (singlet, 1 H), and 2.43 (singlet, 1 H) ppm. What is the structure of D?arrow_forwardCompound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forward
- As we will learn in Chapter 17, reaction of (CH3)2CO with LIC≡CH followed by H2O affords compound D, which has a molecular ion in its mass spectrum at 84 and prominent absorptions in its IR spectrum at 3600−3200, 3303, 2938, and 2120 cm−1. D shows the following 1H NMR spectral data: 1.53 (singlet, 6 H), 2.37 (singlet, 1 H), and 2.43 (singlet, 1 H) ppm. What is the structure of D?arrow_forwardA and B are isomeric dicarbonyl compounds of the molecular formula C5H&O2. The 'H NMR spectrum of A contains a singlet at 2.05 ppm and another singlet at 5.40 ppm. The 'H NMR spectrum of B contains three signals: a singlet at 2.3 ppm, a triplet at 1.10 ppm and a quartet at 2.70 ppm. Suggest structures for A and B and draw them in their respective boxes below. 1st attemptarrow_forwardTreatment of 2-methylpropanenitrile [(CH3)2CHCN] with CH3CH2CH2MgBr, followed by aqueous acid, affords compound V, which has molecular formula C7H14O. V has a strong absorption in its IR spectrum at 1713 cm−1, and gives the following 1H NMR data: 0.91 (triplet, 3 H), 1.09 (doublet, 6 H), 1.6 (multiplet, 2 H), 2.43 (triplet, 2 H), and 2.60 (septet, 1 H) ppm. What is the structure of V? We will learn about this reaction in Chapter 20.arrow_forward
- 1. There are several isomeric alkanes of molecular formula C6H14.Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.84 (d, 12 H), 1.39 (septet, 2H) ppm Isomer B: δ = 0.84 (t, 3 H), 0.86 (s, 9H), 1.22 (q, 2H) ppmarrow_forwardTreatment of benzoic acid (C6H5CO2H) with NaOH followed by 1-iodo-3methylbutane forms H. H has a molecular ion at 192 and IR absorptions at 3064, 3035, 2960−2872, and 1721 cm−1. Propose a structure for H.arrow_forwardReaction of (CH3)3CCHO with (C6H5)3P=C(CH3)OCH3, followed by treatment with aqueous acid, affords R (C7H14O). R has a strong absorption in its IR spectrum at 1717 cm−1 and three singlets in its 1H NMR spectrum at 1.02 (9 H), 2.13 (3 H), and 2.33 (2 H) ppm. What is the structure of R? We will learn about this reaction in Chapter 18.arrow_forward
- G.267.arrow_forwardThere are several isomeric alkanes of molecular formula C6H14. Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.84 (d, 12 H), 1.39 (septet, 2H) ppm Isomer B: δ = 0.84 (t, 3 H), 0.86 (t, 9H), 1.22 (q, 2H) ppmarrow_forwardThe treatment of (CH3)2C=CHCH2Br with H2O forms B (molecular formulaC5H10O) as one of the products. Determine the structure of B from its 1H NMR and IR spectra.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning