1. For each of the following pairs of solute and solvent, predict whether the solute would be soluble or insoluble. After making your predictions, you can check your answers by looking up the compounds in The Merck Index or the CRC Handbook of Chemistry and Physics. Generally, The Merck Index is the easier reference book to use. If the substance has a solubility greater than 40 mg/mL, you conclude that it is soluble. a. Malic acid in water НО -С-СНСН,—С—ОН ÓH Malic acid b. Naphthalene in water Naphthalene c. Amphetamine in ethyl alcohol NH2 -CH2CHCH3 Amphetamine d. Aspirin in water -С-ОН 0-C–CH3 Aspirin
1. For each of the following pairs of solute and solvent, predict whether the solute would be soluble or insoluble. After making your predictions, you can check your answers by looking up the compounds in The Merck Index or the CRC Handbook of Chemistry and Physics. Generally, The Merck Index is the easier reference book to use. If the substance has a solubility greater than 40 mg/mL, you conclude that it is soluble. a. Malic acid in water НО -С-СНСН,—С—ОН ÓH Malic acid b. Naphthalene in water Naphthalene c. Amphetamine in ethyl alcohol NH2 -CH2CHCH3 Amphetamine d. Aspirin in water -С-ОН 0-C–CH3 Aspirin
Chapter1: Solubility
Section: Chapter Questions
Problem 1Q
Related questions
Question
1: Need help with a,b,c,d
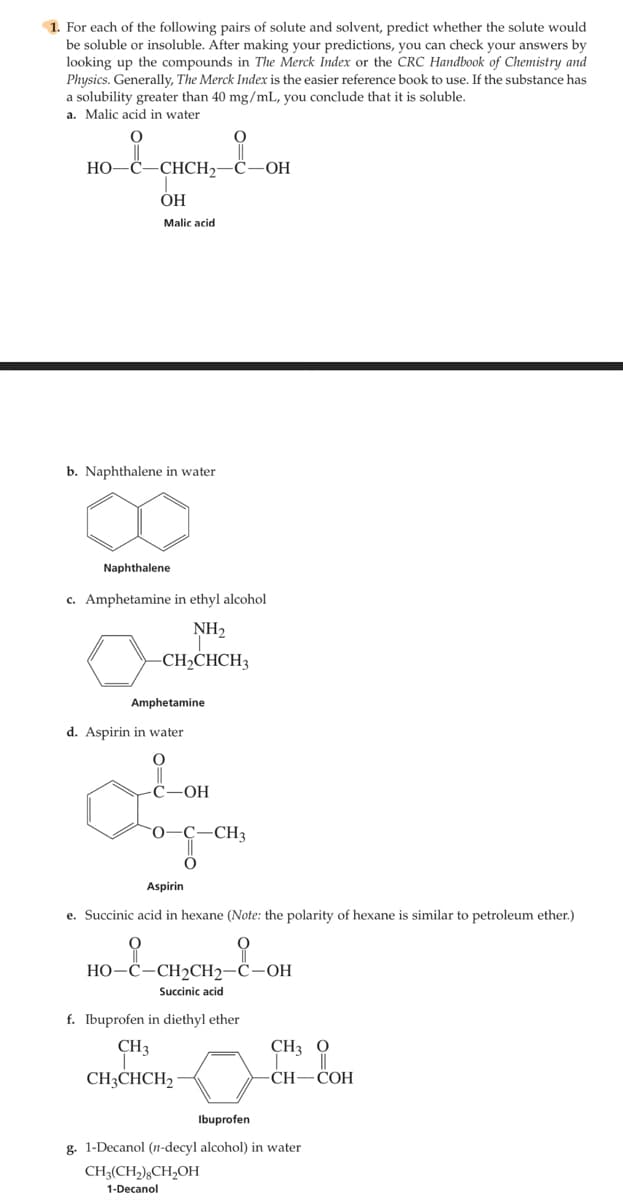
Transcribed Image Text:1. For each of the following pairs of solute and solvent, predict whether the solute would
be soluble or insoluble. After making your predictions, you can check your answers by
looking up the compounds in The Merck Index or the CRC Handbook of Chemistry and
Physics. Generally, The Merck Index is the easier reference book to use. If the substance has
a solubility greater than 40 mg/mL, you conclude that it is soluble,
a. Malic acid in water
HO-
-CHCH2
OH
ОН
Malic acid
b. Naphthalene in water
Naphthalene
c. Amphetamine in ethyl alcohol
NH2
CH»CHCH3
Amphetamine
d. Aspirin in water
С—ОН
0-Ç
-CH3
Aspirin
e. Succinic acid in hexane (Note: the polarity of hexane is similar to petroleum ether.)
НО-С-СН2СH2—С—ОН
Succinic acid
f. Ibuprofen in diethyl ether
CH3
CH3 0
CH;CHCH2 ·
CH—СОН
Ibuprofen
g. 1-Decanol (n-decyl alcohol) in water
CH3(CH,);CH,OH
1-Decanol
Expert Solution
Step 1 Introduction
We have find out the solubility of solute
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning