1. In a serial dilution, one initially sets a starting dilution and adds an aliquot portion of it successively to other tubes. Refer to the table below and fill in the missing values.
1. In a serial dilution, one initially sets a starting dilution and adds an aliquot portion of it successively to other tubes. Refer to the table below and fill in the missing values.
Chapter13: Dimensional Analysis/units Conversion
Section: Chapter Questions
Problem 1.10P
Related questions
Question
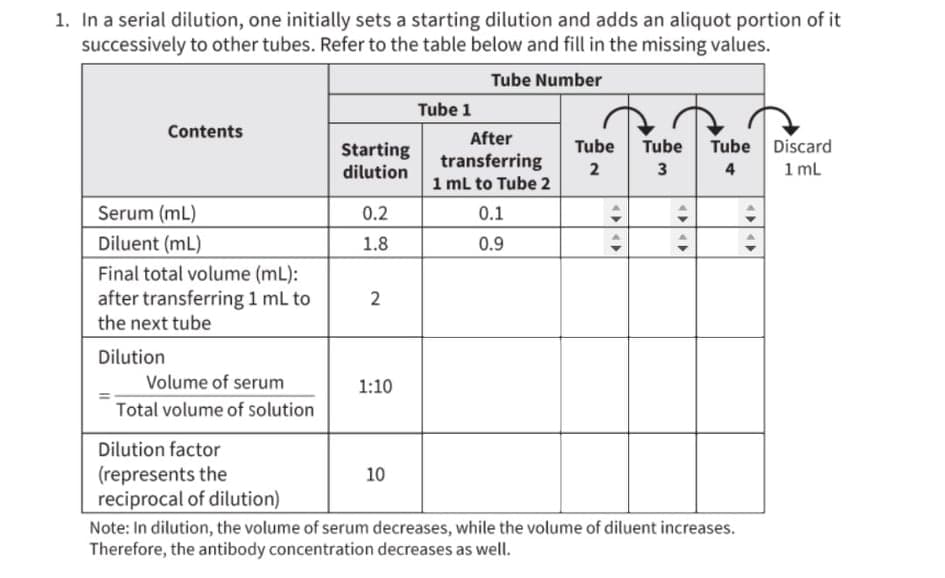
Transcribed Image Text:1. In a serial dilution, one initially sets a starting dilution and adds an aliquot portion of it
successively to other tubes. Refer to the table below and fill in the missing values.
Tube Number
Tube 1
Contents
After
Tube Discard
1 mL
Tube
Tube
Starting
dilution
transferring
1 mL to Tube 2
4
Serum (mL)
0.2
0.1
Diluent (mL)
1.8
0.9
Final total volume (mL):
after transferring 1 mL to
the next tube
2
Dilution
Volume of serum
1:10
Total volume of solution
Dilution factor
(represents the
reciprocal of dilution)
Note: In dilution, the volume of serum decreases, while the volume of diluent increases.
Therefore, the antibody concentration decreases as well.
10
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Cardiopulmonary Anatomy & Physiology
Biology
ISBN:
9781337794909
Author:
Des Jardins, Terry.
Publisher:
Cengage Learning,


Cardiopulmonary Anatomy & Physiology
Biology
ISBN:
9781337794909
Author:
Des Jardins, Terry.
Publisher:
Cengage Learning,