
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
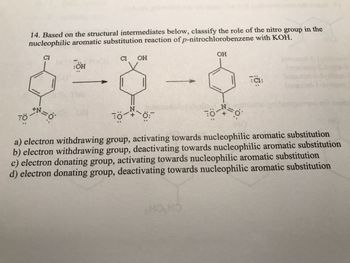
Transcribed Image Text:14. Based on the structural intermediates below, classify the role of the nitro group in the
nucleophilic aromatic substitution reaction of p-nitrochlorobenzene with KOH.
+N
TÖ
C1
:ÖH
C1 OH
<
N
TÖ +
³0:
F
HE
OH
HO
N
70+
a) electron withdrawing group, activating towards nucleophilic aromatic substitution
b) electron withdrawing group, deactivating towards nucleophilic aromatic substitution
c) electron donating group, activating towards nucleophilic aromatic substitution
d) electron donating group, deactivating towards nucleophilic aromatic substitution
TCI:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Could you please help with these questions? Thank you!arrow_forward6. Consider the following reaction scheme: 1) l2, NAOH, H,0 2 OH + ICH3 2 2) H30* Draw a detailed mechanism with arrows showing the movement of electrons for the formation of benzoic acid and iodoform from 1,3-diphenyl-1,3-propanedione (2).arrow_forward+ Module 10 HW Chapter 20 - Selected Exercises Question 3 of 7 -/1 E Draw the major product(s) formed when cyclopentanecarboxylic acid is treated with each of the following reagents: with SOCI2: * Draw Your Solution Q Mu eTextbook and Media Que Fill in with LIAIH, (excess), followed by HạO": Ques Multipa MacBook Air esc 000 F4 トト FI F2 F3 FS F6 F7 %23 %24 % & * 2 4 5 6 8 W E R T Y tab S D F K caps lock C shift alt control option command CO >arrow_forward
- Question 13 of 17 0.67 / 1 View Policies Show Attempt History Current Attempt in Progress Propose a plausible mechanism for the following transformation. 1) NaH OEt 2) Etl 13.38b Correct. This mechanism begins with deprotonation of the alcohol by the strong base. The resulting alkoxide anion is a strong nucleophile and will undergo an SN2 mechanism with ethyl iodide. 2 v This substitution reaction is expected to be an SN iodide mechanism because the leaving group, IIarrow_forwardPlease don't provide handwriting solutionarrow_forwardStereoisomers of α-bromocinnamic acid: draw the reaction mechiansim with arows for the dehydrobromination of 2,3-dibromo-3-phenylpropanoic acid with ethanol and KOH to produce (Z)-α-bromocinnamic acid and (E)-α-bromocinnamic acid. Also draw the transition state for The (Z)-isomer syn-periplanar transition state and anti-periplanar transition state of the (E)-isomer.arrow_forward
- Please help with the following ochem reaction scheme involving ethyl 3-methyl-2-oxocyclopentane-1-carboxylate. Provide the bond line structures for each compound formed in each step of the reaction scheme shown in the image providedarrow_forward17 eBook Print References Specify reagents suitable for converting 3-ethyl-2-pentene to 2,3-epoxy-3-ethylpentane. Select the single best answer. peroxyacetic acid 1. B₂H6; 2. H₂O2, HO™ 3 attempts left Check my work O H₂O, H₂SO4 Cr₂0₂², H 03, CH3OHarrow_forward7. Hoe ga je onderstaande cyclische ether synthetiseren indien je enkel beschikt over 5-bromo-1-penteen, verdund zwavelzuur, NaH, 9-BDN/THF, H₂O₂/OH-/H₂O (je hoeft niet alle materialen te gebruiken)? Geef het volledige reactiemechanisme. : 0arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY