150 75-- t1/2 37.5 t1/2- 10,000 30,000 20,000 Time (s) A Figure 14.10 Kinetic data for the rearrangement of methyl isonitrile to acetonitrile at 199 °C, showing the half-life of the reaction. Pressure, CH3NC (torr)
150 75-- t1/2 37.5 t1/2- 10,000 30,000 20,000 Time (s) A Figure 14.10 Kinetic data for the rearrangement of methyl isonitrile to acetonitrile at 199 °C, showing the half-life of the reaction. Pressure, CH3NC (torr)
Chapter12: Chemical Kinetics
Section: Chapter Questions
Problem 12Q: The plot below shows the number of collisions with a particular energy for two different...
Related questions
Question
If a solution containing 10.0 g of a substance reacts by first-order
kinetics, how many grams remain after three half-lives?
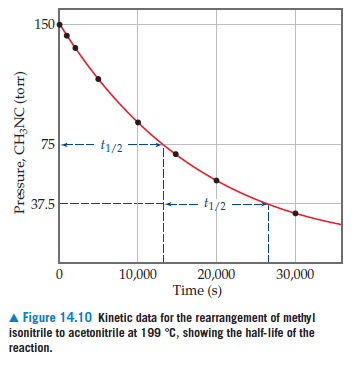
Transcribed Image Text:150
75--
t1/2
37.5
t1/2-
10,000
30,000
20,000
Time (s)
A Figure 14.10 Kinetic data for the rearrangement of methyl
isonitrile to acetonitrile at 199 °C, showing the half-life of the
reaction.
Pressure, CH3NC (torr)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning