Q: Siven reaction is carried out in acidic medium with a yield of 91.5%. How many grams of CH,CHO is…
A:
Q: A stock solution of potassium permanganate (KMn04) was prepared by dissolving 13.0g KMn04 with DI in…
A: Record the given data, The given weight of KMnO4 = 13.0 gm The molecular weight of KMnO4 = 158 g/mol…
Q: a solution that is 0.160 M in propanoic acid and 0.110 M in potassium propanoate (where pK, = 4.87…
A: pH of a buffer is given by the following equation. pH=pKa + log[base][acid]
Q: Calculate the concentration of Pd<t ion in an unknown sample based on the following data: Vol. of…
A: According to the mole concept, in terms of mass, the amount of substance in moles is equal to the…
Q: Q1. Dissolved 0.273 grams of pure sodium oxalate (Na.C.O.) in distilled water and added sulfuric…
A: Normality of solution is equal to equivalents per litre. And Normality = n-factor × Molarity
Q: The %purity of a powdered crude sample of Na2CO3 containing only inert impurities is to be…
A: The balanced chemical equation between Na2CO3 and HCl to produce NaCl, H2O and CO2 can be…
Q: Compare and contrast an anticipatory standard to a reactionary standard by listing and describing…
A: In sole proprietorship type of business organization, you would be fully responsible for all debts…
Q: The weight of 5 ml of vinegar sample was 5.228 g, was required 30.5 ml of 0.2 M sodium hydroxide…
A: Acid reacts with base to form salt and water and this reaction is known as neutralization reaction.…
Q: A chemical factory has been illegally disposing their chemical wastes without necessary…
A:
Q: 1. To complete the following table: a. Determine the initial concentrations (molarity) of S2032 and…
A:
Q: Electrolytic manganese dioxide can be prepared from manganese carbonate ore by crushing, milling and…
A: Preparation of manganese dioxide from magnesium carbonate ore by crushing, milling and leaching the…
Q: what is molarity, the average molarity, and standard deviation? igmsa H Titrations - Report Sheet…
A:
Q: An analyst weighed 0.25045 g of the Buffalo River Sediment reference material. (RM # 8704) to…
A:
Q: 1. A 4.59 mL sample of HCl, specific gravity 1.3, required 50.5 mL of 0.9544N NaOH in a titration.…
A:
Q: (c) The ore being placed on the heap has an average copper grade of 1.0% and the lixiviant addition…
A: The average recovery of the copper from the heap leaching is 90%.
Q: The %purity of a powdered crude sample of Na2CO3 containing only inert impurities is to be…
A: #(a): Na2CO3 reacts with HCl to produce H2CO3(aq) and NaCl. H2CO3(aq) decomposes further to form…
Q: Consider the following method for the analysis of cocaine (MW 303.35) powder. "Dissolve about 600 mg…
A: Given the mass of cocaine powder weighed = 606.3 mg molarity of HClO3 used = 0.09978 M Volume of…
Q: Part I. Cause and Effect: lIdentify the effect of the condition on the indicated parameter. Write…
A: Higher concentration solution will have more mass of solution. Hence low specific heat. With the use…
Q: A ceric sulfate solution that was standardized using 200 mg of arsenic trioxide (MW 197.84g/n)…
A:
Q: 1. The Ksp of Ca3(PO4)2 is 1.3 × 10−26. Estimate the solubility of this salt in units of g. L−1…
A:
Q: Which of the following are equivalent to 2,500 ppm Cu2+? (There may be more than one answer)…
A: Given data 2500 ppm equal to
Q: 2- Equivalent conductance of 1 M propanoic acid CH3CH2COOH 10 ohm cm? eq and that at infinite…
A:
Q: Using a 10mL pipette, you prepare a standard solution by pipetting 10.00mL of the standard iron…
A: A particular pipette has an approximate tolerance or uncertainty value. Based on these uncertainty…
Q: e used to determine whether the concentrations of haloacetates in the treated water demonstrate…
A: Here, the DBPR - two levels. The level 1 of DBPR new standards and a treatment technique of enhanced…
Q: Chemistry An aquifer contaminated with petroleum is found to have the following component…
A: The age of the spill can be evaluated with the help of the BTEX ratio. 'B' implies benzene, 'T'…
Q: To a 0.5 g sample of impure NaCl are added 0.784 g of pure AgNO3 (MW = 169.88) crystal. This is in…
A: A precipitation reaction involves the formation of a solid, insoluble substance. In Stoichiometry,…
Q: Compound Observation H2O 5% NaOH 5% NaHCO3 HCI H2SO4 Hexane insoluble insoluble insoluble insoluble…
A: The above table can be explained on the basis of solubility of like dissolves like. Ionic compounds…
Q: and in 4) To adjust the thiosulfate (S2O32) solution to be used as a titrant in the copper…
A:
Q: A 0.1475-M solution of Ba(OH)2 was used to titrate the acetic acid (60.05 g/mol) in a dilute aqueous…
A: In order to find the mean w/v percentage of acetic acid in the sample. we have to find the w/v…
Q: 0.5M HA sample with 0.2 dissociation; It is placed in a conductivity cell with a length of 0.9 cm…
A: Given: Concentration of weak electrolyte = 0.5 M Length = 0.9 cm Area = 2cm2
Q: You have Falcon tubes containing 10 ml and you should add ,that you have in a voltage series, from…
A:
Q: A ceric sulfate solution that was standardized using 200 mg of arsenic trioxide (MW = 197.84g/n)…
A:
Q: Soil Fertility Index and Soil Evaluation Factors, Are these equations suitable for alkaline soil?…
A: Soil is a combination of organic matter, rocks, gases, liquids and organisms that sustain life…
Q: < E session.masteringchemistry.com MasteringChemistry: HW 8 <HW 8 Applications for Dilution…
A: From the given data in the question, sayThe final concentration of the diluted solution = cVolume…
Q: Calculate the solubility at 25 °C of PbCO3 in pure water and in a 0.0070M Pb(NO3), solution. You'll…
A:
Q: Mganga is doing a routine analysis in a copper plating factory. He is regularly making up solutions…
A: The number of moles is a ratio of the mass of a substance to the molar mass of the substance. 'N' is…
Q: Cg 162 liters of a liquid chemical with a dimension less Henry's volatility constant of KG° Caq…
A:
Q: The student then determined ΔH neutralization for the reaction of sodium hydroxide and acetic acid,…
A: First calculate moles of each reactant by multiply MOLARITY and volume then find out limiting…
Q: The %purity of a powdered crude sample of Na2CO3 containing only inert impurities is to be…
A: A eudiometer is used to find the volume of a gas produced during a chemical reaction. It is also…
Q: Consider the following method for the analysis of cocaine (MW 303.35) powder. "Dissolve about 600 mg…
A: For acid base titartion: Nacid x Vacid = Nbase x Vbase Since the given acid is monoprotic and there…
Q: eloom СЗ. Concentration of NH4CI solution: Calculated pK, of NH4*: (show calculations)
A:
Q: A water containing 50 mg/L of phosphate (PO.) is flowing in an open channel at a volumetric flow…
A: One liter of water contains 50mg of phosphate ions and the total liter of water flowing per day is…
Q: Ksp=4.1x10-36 for Pb3(AsO4)2 Calculate Eo for Pb3(AsO4)2(s) + 6e- 3Pb(s) + 2AsO4 3- Pb2+ +2e- …
A: Given data: KSP =4.1x10-36 E0pb2+/pb = -0.126V Number of electrons = 6 e-
Q: Calculate the solubility at 25 °C of PbCO3 in pure water and in a 0.0100M Pb(NO3), solution. You'll…
A:
Q: Gravimetric Problems A 516.7-mg sample containing a mixture of K,SO, and (NH,)SO, was dissolved in…
A: Given, Mass of sample: 516.7 mg=516.7×10−3 g BaSO4 yield = 863.5 mg The balanced chemical equation:…
Q: Calculate the solubility at 25 °C of PbCrO4 in pure water and in a 0.0080M Na₂ CrO4 solution. You'll…
A: #(1): Calculation for solubility in pure water: Ksp of PbCrO4 = 2.8*10-13
Q: Standard Solutions for FENCS2* Ion Beer-Lambert Plot mL of 2.00 x 101 M Fe(NO,)3 in 0.10 M HNO, mL…
A:
Q: c. An Analytical laboratory at SMU has a bottle of concentrated HCI solution labelled, specific…
A: C). given that Specific gravity = 1.2 g/cm3 the density of the solution will be equal to the…
do all the following listed on the second photo (bullet) to the nernst equation given. thank you
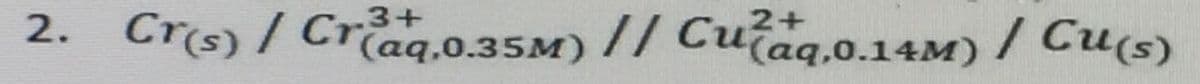

Step by step
Solved in 4 steps

- What is the Ksp of Zn3(PO4)2 (MW = 320.8 g/mol) if its solubility in water at 25 °C is 4.9 x 10-5 g/L? format for the scientifc notation 1.0 x 10^-1avg concentration: 8.937x10^-7A spectrophotometric method for the quantitative analysis of Pb2+ in blood has a normal calibration curve for which Sstd=(0.296 ppb−1)×Cstd+0.003. What is the concentration of Pb2+ in a sample of blood if Ssamp is 0.397?
- A titrimetric method for the determination of calcium in limestone was tested by analysis of an NIST limestone containing 30.15% CaO. The mean result of four analyses was 30.26% CaO, with a standard deviation of 0.085%. By pooling data from several analyses, it was established that s = 0.094% CaO.a. Do the data indicate the presence of a systematic error at the 95% confidence level?b. Do the data indicate the presence of systematic error at the 95% confidence level if no pooled value for s was available?You are developing a Ca detection method for a graphite furnace AA instrument. Your analysis of ten blank samples gives a standard deviation of 0.265 ppb. To establish your limit of detection (LOD), what concentration of Ca should be spiked into your blanks?(9.29 × 105/8)-20.81 = with correct sig figs
- Concentration=2.5 X 10^3 μM OD400nm=0.859 1cm cuvette is used what is the extinction coefficient (μM^-1 cm^-1)Calculate the [FeSCN2+] using volumes of stock solutions. Presume that all the SCN– ions react. STANDARD SAMPLE Volume of Fe(NO3)3 (mL) Volume of SCN- (mL) Volume of H2O (mL) [FeSCN2+] Absorbance 1 2.5 2.0 20.5 0.1918 2 2.5 1.5 21.0 0.3239 3 2.5 1.0 21.5 0.4965 4 2.5 0.5 22.0 0.6209 Stock [Fe(NO3)3] = 0.200 M, Stock [SCN-] = 0.0020 M Any help would be greatly appreciated Please and thanks :)Find (M) for [FeSCN^2+] using the following info: 0.200 M (Iron (III) nitrate) Fe(NO3)3 (mL) = 17.98 mL 0.00200 M (Potassium thiocyanate) KSCN (mL) = 1.97 mL 0.815 Absorbance
- In this experiment you will be preparing aqueous solutions of [Co(NH3)5N3]Cl2 at fixed concentrations. Given Mr ([Co(NH3)5N3]Cl2) = 257.01, what mass of [Co(NH3)5N3]Cl2 is required to prepare 10 mL of 0.0200 mol.dm-3 solution?Explain why Fe3+ is in large excess in the standardization procedure. Fe3+ + SCN- → FeNCS2+Elaborat ur answer pls thank u

