Q: Wolff-Kishner reduction of compound W gave compound A. Treatment of A with m-chloroperbenzoic acid…
A: Given is, Wolff-Kishner reduction of compound W gave compound A. Treatment of A with…
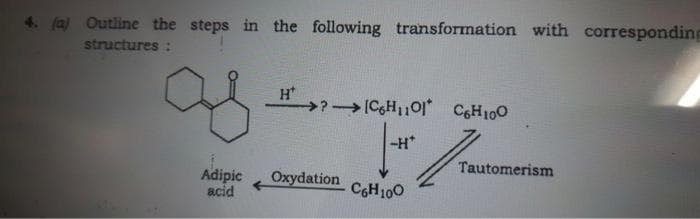
Q: Using the normal/primary isomer of C4H10O, treat it with conc.H2SO4 and heat to produce A.…
A: The primary isomer of C4H10O must be a primary alcohol thus, C4H10O is butan-1-ol. The structure of…
Q: (d) For the following reaction give the structure of the heterocyclic product formed and give a…
A: This reaction is an example of Heck Coupling.
Q: Butanone is formed when an alkyne is passed through a dil sol of H2SO4at 330K in presence of…
A: Given: Butanone is formed when an alkyne is passed through a dil sol of H2SO4 at 330K in presence of…
Q: 2) Provide the structure of the major organic product of the following reaction. O HN C₂H5COCI AIC13
A:
Q: and HO, ÓH
A: Given products from the reaction,
Q: 3) Isoerythrogenic acid, C18H2602, is an acetylic fatty acid that turns a vivid blue on exposure to…
A: Based on the data given in above question, we can draw the line structure as below- This is also…
Q: 3. Isoerythrogenic acid, C18H26O2, is an acetylenic fatty acid that turns a vivid blue on exposure…
A:
Q: 0. H3C H3C H+ Propanol H+ ? 2
A:
Q: synthesis
A:
Q: 4. Compound CC is formed from the reaction of benzene with CH3;Cl in the presence of AICI3.…
A: (a. The structural formula of CC, EE, FF, GG and DD can be given as follows:…
Q: An unknown alcohol C6H14O gave a rapid Lucas Test. It could not be oxidized easily, but upon…
A: DBE of the alcohol = (C+1)-H/2 = (6+1)-14/2 = 0. Hence, it is an aliphatic alcohol. According to…
Q: 3. Write in the product of 3-isobutyl-1-heptyne with the following reagents: 1. ВНз 2. Н,Ог, "ОН 1)…
A:
Q: 4. Predict the biotransformation and conjugation metabolite of below molecule
A: Biotransformation is the alteration of molecule without breaking the bonds.
Q: Give a proposal on the synthesis methods of CoSO4.7H2O
A: Cobalt (II) sulphate can be synthesized by the reaction of cobalt metal with aqueous solution of…
Q: (E)-1-2- Diphenylethene reacts with Br 2 /CCI4 (see reaction) the reaction product is obtained (A)…
A:
Q: 1.When 2-heptyne was treated with H2SO4(aqueous) containing mercuric sulphate, two products, each…
A: This is an example of oxymercuration demercuration reaction. Alkynes undergo this reaction to form…
Q: Hydrocarbon A has the formula C9H12 and absorbs 3 equivalents of H2 to yield B, C9H18, when…
A:
Q: Which of the alkenes below is the most reactive towards acid-catalyzed hydration?
A: Markovnikovs rule is applied to acid catalysed hydration of alkene.
Q: Acid-catalyzed hydrolysis of the following epoxide gives a trans diol. CH3 CH3 CH3 НО, НО, H,SO, +…
A: This reaction proceeds by an acid-catalyzed ring opening of the epoxide with water acting as a…
Q: Outline the synthesis of racemic 3-methyl-3-heptanol, Et(Me)C(*)OHn-Bu, starting from alcohols of…
A: Using a 4 or less membered alcohol, we have to form 3-methyl 3-heptanol.
Q: Compound A has the formula CioH16 On catalytic hydrogenation over palladium, it reacts with only one…
A: Ozonolysis is an organic reaction shown by unsaturated alkenes and alkynes. The carbon-carbon…
Q: One of the constituents of turpentine is a-pinene, formula C10H16. The following scheme gives some…
A:
Q: Helenalin is a terpene that can be isolated in several plants such as Arnica montand alU A ICU…
A: Terpene: Helenalin
Q: Determine a possible structure for an alkene, X, formula C9H14, on the basis of the following…
A: Reaction involved ( given) are :- 1. Catalytic hydrogenation :- Alkene get converted to alkane due…
Q: 5. Provide all the missing products (A-C) and reagent (i). Suggest retrosynthetic analysis for this.…
A:
Q: 6.5 Outline the synthesis of 4-methyl-2-pentanone from acetoacetic ester, benzene, and alcohols of…
A:
Q: 4.- Provide an example of the use of (S)-proline as organocatalyst, and mention how this catalyst…
A:
Q: The compound BD3 is a deuterated form of borane. Predict the product formed when 1-methylcyclohexene…
A: Given compound: 1-methylcyclohexene
Q: Deduce the structure of each compound from the information given. All unknowns in this problem have…
A: The molecular formula C8H12 suggests three elements of unsaturation. Catalytic hydrogenation gives…
Q: 10. MM and NN are amines with molecular formula, C3H9N. Reaction of MM with sodium nitrite and HCl…
A: Amine react with nitrous acid form different compound
Q: Et CH,OH, H* H2. Ni Raney H,0, H* Me Chauffage Me
A:
Q: 2 steps BU4NF C9H1602 A + B ÓSIME,tBu (i) Enone C can be prepared from alkyne A and aldehyde B in…
A:
Q: Q2. Outline the possible reaction pathways with propose mechanism for the formation of Nylon 6 from…
A: Polymer can be defined as the chemical compound that is formed by the combination of one or more…
Q: CHs Н,с Но,д осн,сн, н-сн,cо Complete the following reaction by filling in the necessary reagents. 6…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Compound A, C14H10, absorbed only 2 equivalents of H2 on catalytic hydrogenation over a Pd/C…
A:
Q: 2. The carboxyl group on a benzene ring is a(n) (ortho-para or meta) director in electrophilic…
A:
Q: An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a…
A: Reduction of alkene gives alkane.
Q: A compound A, C7H12, was found to be optically active. On catalytic reduction over platinum…
A: Alkynes are organic compounds containing carbon and hydrogen atoms. In alkynes, there is a presence…
Q: Isoerythrogenic acid, C18H26O2, is an acetylic fatty acid that turns a vivid blue on exposure to UV…
A: Based on the data given above, the line structure is shown below- It is also known as isanic acid.
Q: When A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With…
A: The question is based on the concept of organic reactions. We have to identify the product and…
Q: Ribose, a carbohydrate with the formula shown, forms a cyclic hemiacetal, which, in principle, could…
A: Ribose is the carbohydrates or in a simple form, they are sugar having molecular formula C5H10O5.…
Q: (d) Assuming you also have available BD3:THF and CH3CO2T, can you suggest a synthesis of the…
A: Hydroboration-oxidation is the addition of borane across the double bond followed by oxidation using…
Q: Chemical compound A is an alkene that when treated with ozone followed by treatment of Zn in HOAc…
A:
Q: b. Propose an electron pushing mechanism for the cyclization of the amide intermediate to…
A: The solution is given below -
Q: A chiral amine A having the R conguration undergoes Hofmann elimination to form an alkene B as the…
A: The ozonolysis of an alkene using reductive work up cleaves double bond to form two aldehyde or two…
Q: -s-OH Methanesulfonic acid Isopropyl cyclopentanecarboxylate H20 ,A
A:
Step by step
Solved in 3 steps with 2 images

- Friedel–Crafts alkylation of benzene with (R)-2-chlorobutane and AlCl3 affords sec-butylbenzene.a. How many stereogenic centers are present in the product?b. Would you expect the product to exhibit optical activity? Explain, with reference to the mechanism.Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d belowThe (+) - aureol is a natural product that has shown selective activity against certain colon and lung cancer cell lines. It is postulated that a key step in the biosynthesis of this substance is the conversion of carbocation A into B. Propose a possible mechanism for this transformation (MUST show the corresponding arrows).
- 4. Give the structure of compounds A to E in the following reaction sequence.The last step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass found in tropical and subtropical regions of the world, involves treatment of C with CH3Li to form an intermediate X, which forms β-vetivone with aqueous acid. Identify the structure of X and draw a mechanism for converting X to β-vetivone.Provide a mechanism for the following isomerization in aqueous acid. Thank you in advance!
- Write out all the isomers of the compound with molecular formula C4H10O . Select the normal/ primaryisomer and treat it with conc. H2SO4 and heat. Identify the reaction and give the product ‘A’ from it. 2. When ‘A’ is treated with HBr in the presence of a peroxide, give the name and structure of the product.The reaction proceeds by an SN2 mechanism. Draw a sketch of the structure of the transitionstate, clearly indicating:- the geometry at the electrophilic carbon centre, and- bonds in the process of being broken or formed with dotted lines, if present.Doxaprost, an orally active bronchodilator patterned after the natural prostaglandins , is synthesized in the following series of reactions starting with ethyl 2-oxocyclopentanecarboxylate. Except for the Nef reaction in Step 8, we have seen examples of all other types of reactions involved in this synthesis. Q. Describe experimental conditions to bring about Step 7 and account for the fact that the trans isomer is formed in this step.
- Compound A (C9H12) absorbed 3 equivalents of H2 on catalytic reduction over apalladium catalyst to give B (C9H18). On ozonolysis, compound A produced 3 products, C, Dand E which was a ketone identified as cyclohexanone. On treatment with NaNH2 in NH3,followed by addition of iodomethane, compound A gave new hydrocarbon, F (C10H14). Drawstructures of A, B, C, D and F.A certain hydrocarbon, C7H10, yields the two products shown below upon oxidative cleavage with KMnO4/H3O+ . Deduce the structure of the hydrocarbon.(S)-2-Butanol, CH3CH2CHOHCH3, slowly racemizes on standing in dilute aqueous sulfuric acid. Explain, illustrating with a mechanism.

